Does copper have any isotopes
Natural copper comprises two stable isotopes, 63Cu (69.17%) and 65Cu (30.83%), and 27 known radioisotopes, five of them are particularly interesting for molecular imaging applications (60Cu, 61Cu, 62Cu, and 64Cu), and in vivo targeted radiation therapy (64Cu and 67Cu) [4].
How many isotopes of copper are there?
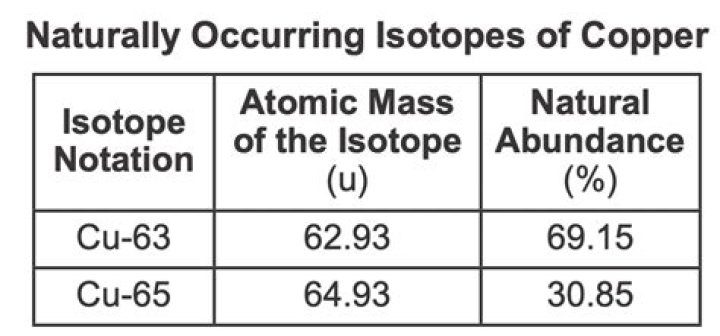
Copper has two stable isotopes, 63Cu and 65Cu, with relative abundances of 69.15 % and 30.85 %, respectively. A transition metal, Cu is moderately siderophile and strongly chalcophile (around 2/3 of Earth’s Cu is thought to be stored in its core).
Is Copper 65 an isotope?
Copper 65 Metal (Copper-65) is a stable (non-radioactive) isotope of Copper. It is both naturally occurring and a produced by fission. Copper 65 Metal is one of over 250 stable Metallic isotopes produced by American Elements for biological and biomedical labeling, as target materials and other applications.
What are the 2 copper isotopes?
Copper has two naturally occurring isotopes, 63Cu (62.9396 amu) and 65Cu (64.9278 amu). If copper has an atomic mass of 63.546 amu, what is the percent abundance of each copper isotope?What are the isotopes of copper used for?
Both isotopes of Copper, Cu-63 and Cu-65, are used to study copper metabolism and gastrointestinal diseases. Cu-63 is used for production of medical radioisotope Zn-62 and can also be used for the production of Cu-64 which is used in cancer diagnosis and treatment.
How many isotopes does copper 63 have?
So, you know that copper has two naturally occurring isotopes, copper-63 and copper-65. This means that their respective decimal abundance must add up to give 1 . If you take x to be the decimal abundance of copper-63, you can say that the decimal abundance of copper-65 will be equal to 1−x .
How do you determine isotopes?
Subtract the atomic number (the number of protons) from the rounded atomic weight. This gives you the number of neutrons in the most common isotope. Use the interactive periodic table at The Berkeley Laboratory Isotopes Project to find what other isotopes of that element exist.
How many isotopes does zinc have?
With the middling atomic number 30, it has five stable isotopes of atomic weight from the dominant zinc 64 to zinc 70, plus an extra 25 radioisotopes.How many isotopes does Sn have?
Tin has 10 stable isotopes, occurring in the following percentages in natural tin: tin-112, 0.97; tin-114, 0.65; tin-115, 0.36; tin-116, 14.53; tin-117, 7.68; tin-118, 24.22; tin-119, 8.58; tin-120, 32.59; tin-122, 4.63; and tin-124, 5.79.
How many isotopes does copper 65 have?1 Physical and Chemical Properties Copper has two stable isotopes, 63Cu and 65Cu, with natural abundances of 69.2% and 30.8%, respectively (Georgopoulos et al., 2001). The water solubility of copper(II) sulfate (Chemical Abstracts Service (CAS) No. 7758-98-7) is 143 g/L at 0°C, whereas cuprous(I) oxide (CAS No.
Article first time published onIs copper a synthetic?
Copper metal does occur naturally, but by far the greatest source is in minerals such as chalcopyrite and bornite. Copper is obtained from these ores and minerals by smelting, leaching and electrolysis. The major copper-producing countries are Chile, Peru and China.
Is cobalt 59 an isotope?
The only stable isotope of cobalt is Co-59. But radioactive cobalt can occur naturally, too.
What are three interesting facts about copper?
- Copper has a reddish-metallic coloring that is unique among all the elements. …
- Copper was the first metal to be worked by man, along with gold and meteoritic iron. …
- Copper is an essential element for human nutrition. …
- Copper readily forms alloys with other metals. …
- Copper is a natural antibacterial agent.
Is copper a metal or nonmetal or metalloid?
MetalsNon-metalsMetalloidsCopperHydrogenArsenicIronNitrogenAntimonyMercurySulphurGermaniumZincPhosphorus
What are 3 examples of isotopes?
For example, carbon-12, carbon-13, and carbon-14 are three isotopes of the element carbon with mass numbers 12, 13, and 14, respectively. The atomic number of carbon is 6, which means that every carbon atom has 6 protons so that the neutron numbers of these isotopes are 6, 7, and 8 respectively.
How do you find the most common isotope?
The most common isotope can be found by rounding the atomic weight found on the periodic table of elements to the nearest whole number.
What are 2 examples of isotopes?
Examples of radioactive isotopes include carbon-14, tritium (hydrogen-3), chlorine-36, uranium-235, and uranium-238. Some isotopes are known to have extremely long half-lives (in the order of hundreds of millions of years). Such isotopes are commonly referred to as stable nuclides or stable isotopes.
What is the isotope symbol for copper?
PubChem CID42626467DescriptionCopper-63 is the stable isotope of copper with relative atomic mass 62.929601, 69.2 atom percent natural abundance and nuclear spin 3/2. ChEBI A heavy metal trace element with the atomic symbol Cu, atomic number 29, and atomic weight 63.55. Medical Subject Headings (MeSH)
How many isotopes are shown for copper and what are the abundances of each?
Copper has two isotopes, 63Cu (69.15%, mass=62.9300 amu) and 65Cu (30.85%, mass = 64.928 amu), and so the respective mole fractions are 0.6915 and 0.3085, resulting in an average atomic weight of 63.55 amu, even though there is not a single atom that weighs 63.55 amu.
How many isotopes does antimony have?
Antimony (51Sb) occurs in two stable isotopes, 121Sb and 123Sb. There are 35 artificial radioactive isotopes, the longest-lived of which are 125Sb, with a half-life of 2.75856 years; 124Sb, with a half-life of 60.2 days; and 126Sb, with a half-life of 12.35 days.
How many isotopes does cadmium have?
Cadmium has six isotopes 110, 111, 112, 113, 114, 116.” The isotope 117Cd was first correctly identified by Goldhaber et al.
Is Tin 100 an isotope?
Twenty-nine additional unstable isotopes are known, including the “doubly magic” tin-100 (100Sn) (discovered in 1994) and tin-132 (132Sn). The longest-lived radioisotope is 126Sn, with a half-life of 230,000 years. The other 28 radioisotopes have half-lives less than a year.
How many stable isotopes does neodymium have?
Neodymium is a rare earth element (REE) that occurs in many silicate, phosphate, and carbonate minerals by substitution for major ions. It has seven stable isotopes.
How many isotopes does Mercury have?
Mercury (Hg) is a photochemically active, redox-sensitive metal and exists as multiple physical states in the environment1. It has seven natural stable isotopes (196, 198, 199, 200, 201, 202 and 204) with a relative mass span of 4%.
What are the isotopes of lithium?
Lithium has two stable isotopes Li-6 and Li-7, the latter being 92.5% in nature (hence relative atomic mass of natural lithium of 6.94).
Is zinc a isotope?
Zinc has five stable isotopes; 64Zn (48.63%), 66Zn (27.9%), 67Zn (4.1%), 68Zn (18.75%) and 70Zn (0.62%) (Rosman and Taylor, 1998).
Is copper a mineral or metal?
Copper is a mineral and an element essential to our everyday lives. It is a major industrial metal because of its high ductility, malleability, thermal and electrical conductivity and resistance to corrosion.
Is copper a sulfate?
Copper sulfate is an inorganic compound that combines sulfur with copper. It can kill bacteria, algae, roots, plants, snails, and fungi. … Copper is an essential mineral. It can be found in the environment, foods, and water.
How is copper made naturally?
Copper is a metal that has been deposited from hot sulphur solutions, created in volcanic regions. The hot solutions concentrated the copper up to a thousand times more than would normally be found in rocks. The resultant enriched rocks are called copper ores.
Do all elements have isotopes?
All elements have isotopes. There are two main types of isotopes: stable and unstable (radioactive). … Some elements can only exist in an unstable form (for example, uranium). Hydrogen is the only element whose isotopes have unique names: deuterium for hydrogen with one neutron and tritium for hydrogen with two neutrons.
What are not isotopes?
These elements are vanadium, rubidium, indium, lanthanum, europium, rhenium and lutetium. … In 2 additional cases (bismuth and protactinium), mononuclidic elements occur primordially which are not monoisotopic because the naturally occurring nuclide is radioactive, and thus the element has no stable isotopes at all.