Does cyclohexanone dissolve in water?
Does cyclohexanone dissolve in water?
Water
Cyclohexanone/Soluble in
Why is cyclohexanone soluble in water?
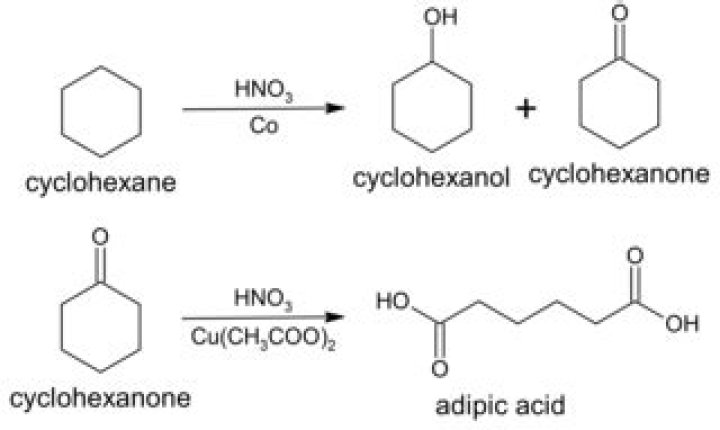
Cyclohexanone is slightly soluble in water and miscible with common organic solvents. Billions of kilograms are produced annually, mainly as a precursor to nylon….Cyclohexanone.
| Names | |
|---|---|
| Melting point | −47 °C (−53 °F; 226 K) |
| Boiling point | 155.65 °C (312.17 °F; 428.80 K) |
| Solubility in water | 8.6 g/100 mL (20 °C) |
What is the structure of Cyclopentanone?
A cyclic ketone that consists of cyclopentane bearing a single oxo substituent. Cyclopentanone is the organic compound with the formula (CH2)4CO. This cyclic ketone is a colorless volatile liquid.
Is cyclohexane soluble?
Cyclohexane is a colourless, mobile liquid with a mild, sweet odour. It is slightly soluble in water and soluble in alcohol, acetone, benzene, ethanol, ethyl ether, olive oil, and carbon tetrachloride.
Is cyclohexanone soluble in ether?
Chemical properties Cyclohexanone is a colorless, clear liquid with soil smell; its impure product appears as light yellow color. It is miscible with several other solvents. easily soluble in ethanol and ether.
Is cyclohexanone polar or non polar?
Solvent Polarity Table
| Solvent | formula | relative polarity2 |
|---|---|---|
| chloroform | CHCl3 | 0.259 |
| cyclohexane | C6H12 | 0.006 |
| cyclohexanol | C6H12O | 0.509 |
| cyclohexanone | C6H10O | 0.281 |
Is Cyclopentanone a ketone?
Cyclopentanone belongs to the class of organic compounds known as ketones. Cyclopentanone is a cyclic ketone, structurally similar to cyclopentane, consisting of a five-membered ring containing a ketone functional group.
What is Cyclopentanone used for?
Cyclopentanone is used as synthesis intermediate for Perfumery products and aromas, Agrochemicals, Pharmaceuticals.