Does ethanol react with Br2?
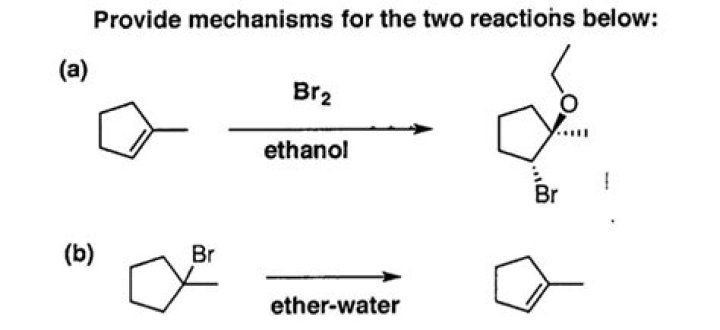
Does ethanol react with Br2?
alcohol (by weight) and bromine in a molar ratio of two to one react at 25°, ethyl acetate and hy- drobromic acid are the sole reaction products. respect to free bromine. The tribromide ion formed during the reaction does not react with alcohol.
What happens when ethanol reacts with bromine?
With Red phosphorus and bromine, ethanol gives ethyl bromide. Acidified potassium dichromate ethyl alcohol gives acetaldehyde and acetic acid.
What chemical formula is Br2?
Br2
Diatomic Bromine/Formula
What is the product when fire on reacts with Br2 in ethanol?
What is the product when pyrrole reacts with Br2 in ethanol? Explanation: This is electrophilic substitution reaction, bromination of pyrrole in presence of ethanol leads to formation of 2,3,4,5-tetrabromopyrrole.
What happens when Ethanoic acid is treated with br2 red phosphorus?
In this reaction red-P and bromine is treated with carboxylic acids having alpha hydrogen atom followed by hydrolysis to form alpha bromo carboxylic acid. Initially red phosphorus and bromine reacts with each other to give PBr₃.
What is Victor Meyer test for alcohols?
Victor Meyer’s Test is one of the prominent tests to distinguish between primary, secondary and tertiary alcohol. In this test, the unknown alcohol is subjected to a series of chemical analysis resulting in observation of colour. The primary alcohol gives blood red colour. The secondary alcohol gives the blue colour.
What is the action of socl2 on ethanol?
Thionyl chloride reacts with ethanol to produce ethyl chloride and hydrogen chloride sulphur dioxide.
What happens when Ethanoic acid is treated with Br2 red phosphorus?
What is Br2 called?
Bromine | Br2 – PubChem.
Is Br2 a element?
Bromine is a chemical element with the symbol Br and atomic number 35….
| Bromine | |
|---|---|
| Phase at STP | liquid |
| Melting point | (Br2) 265.8 K (−7.2 °C, 19 °F) |
| Boiling point | (Br2) 332.0 K (58.8 °C, 137.8 °F) |
What is the product when thiophene reacts with br2 in ch3cooh?
Answer: This is electrophilic substitution reaction, bromination of thiophene in presence of benzene leads to formation of 2,5-dibromothiophene.
What does PBr do to an alcohol?
PBr3 evolves corrosive HBr, which is toxic, and reacts violently with water and alcohols. In reactions that produce phosphorous acid as a by-product, when working up by distillation be aware that this can decompose above about 160 °C to give phosphine which can cause explosions in contact with air.
What is the molar mass of Br2?
Molar mass of Br2 is 159.8080 g/mol Compound name is bromine Convert between Br2 weight and moles
What is the molecular formula for bromoethane?
Bromoethane PubChem CID 6332 Structure Find Similar Structures Chemical Safety Laboratory Chemical Safety Summary (LCSS Molecular Formula C2H5Br or CH3CH2Br Synonyms Bromoethane 74-96-4 ETHYL BROMIDE Ethane
What is the formula for bromine Dibromine?
Bromine PubChem CID 24408 Structure Find Similar Structures Chemical Safety Laboratory Chemical Safety Summary (LCSS Molecular Formula Br2 Synonyms 7726-95-6 Br Bromine Dibromine Brom More
What is the formula for ethyl alcohol?
Ethanol PubChem CID 702 Structure Find Similar Structures Chemical Safety Laboratory Chemical Safety Summary (LCSS Molecular Formula C2H6O or CH3CH2OH Synonyms ethanol ethyl alcohol alcohol 64-17-5 ..