Does heat engine increase entropy?
Does heat engine increase entropy?
Any heat engine operating between the same two temperatures whose efficiency is less than eC necessarily increases the entropy of the universe; in particular, the total entropy of the reservoirs must increase. This increase in entropy of the reservoirs is called entropy generation.
What is the change in entropy of the engine over a full cycle?
When a reversible engine goes through a complete cycle, its change in entropy is zero, as we’ve discussed, and the change in entropy of its environment, that is, the two reservoirs (hot and cold) taken together is also zero: entropy is simply transferred from one to the other.
Does entropy increase in Carnot cycle?
In order to approach the Carnot efficiency, the processes involved in the heat engine cycle must be reversible and involve no change in entropy. This means that the Carnot cycle is an idealization, since no real engine processes are reversible and all real physical processes involve some increase in entropy.
What is the change in entropy for a thermodynamic cycle?
Well-known thermodynamic cycles
| Cycle | Compression, 1→2 | Notes |
|---|---|---|
| Power cycles normally with external combustion – or heat pump cycles: | ||
| Scuderi | adiabatic | |
| Stirling | isothermal | Stirling engines |
| Manson | isothermal | Manson and Manson-Guise engines |
What is entropy vs enthalpy?
Enthalpy is the amount of internal energy contained in a compound whereas entropy is the amount of intrinsic disorder within the compound.
Why is entropy always increasing?
Even though living things are highly ordered and maintain a state of low entropy, the entropy of the universe in total is constantly increasing due to the loss of usable energy with each energy transfer that occurs.
Does entropy change to zero?
The entropy change of the system is given by the difference between the entropy at the final state and the entropy at the initial state. The entropy change is zero for a reversible process.
When heat is added to the system the entropy change is positive?
Defining Entropy and Looking at Entropy Changes in a System If the entropy of a system increases, ΔS is positive. If the entropy of a system decreases, ΔS is negative.
What does entropy change depend on?
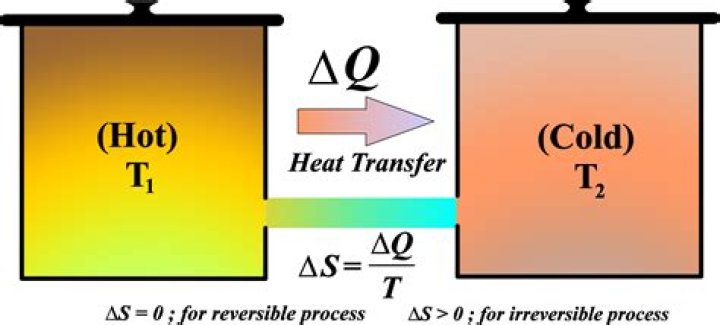
Entropy is given by the equation – delta(S) = Q/T. Therefore Entropy change depends on a greater loss of heat exchange (From Hot to Cold) for a given temperature. A system that is not in equilibrium, or two or more systems that are not in equilibrium.
Why is entropy of Carnot cycle zero?
For a Carnot cycle the entropy change is zero: A S = 0. This is certainly true for the engine because it returns to its initial state after 1 cycle, and entropy is solely a function of the state. During the 2 adiabatic parts of the Carnot cycle, Q = 0, so A S = 0 during the adiabatic processes.
What is the difference between a thermodynamic process and thermodynamic cycle?
In thermodynamics, a “process” is defined by a trajectory in thermodynamic state space from a starting point A to the end point B, and a “cycle” is just a closed trajectory going back to A. Now, there is freedom of choice of the in-between states that the system undergoes.
What is the difference between enthalpy and heat?
What is the difference between heat and enthalpy? Heat is always the energy in transit, i.e, the energy which ‘crosses’ the system boundaries. Whereas Enthalpy refers to total heat content in a system.