How anion is produced?
How anion is produced?
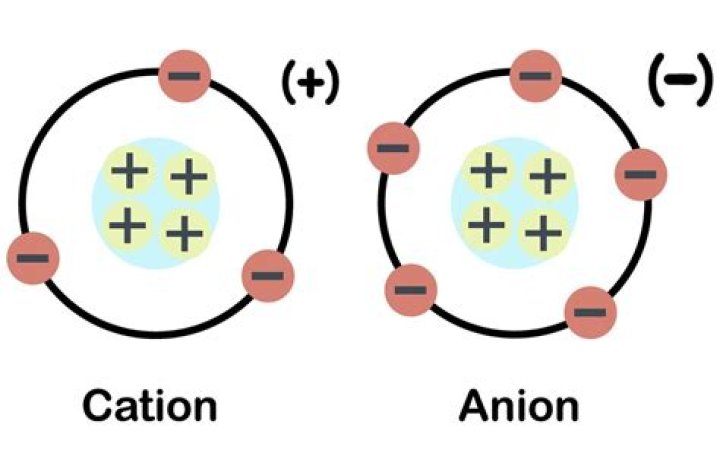
Anions are the negative ions formed from the gain of one or more electrons. When nonmetal atoms gain electrons, they often do so until their outermost principal energy level achieves an octet. Under typical conditions, three electrons is the maximum that will be gained in the formation of anions.
How and why does a anion form?
Anions are negative ions that are formed when a nonmetal atom gains one or more electrons. Anions are so named because they are attracted to the anode (positive field) in an electric field. Atoms typically gain electrons so that they will have the electron configuration of a noble gas.
How are anions formed quizlet?
An atoms loss of valence electrons produces a cation, or a positively charged ion. How do anions form? The gain of negatively charged electrons by a neutral atom produces an anion. Although they are composed of ions,ionic compounds are electrically neutral.
What causes an anion to form?
Cations (positively-charged ions) and anions (negatively-charged ions) are formed when a metal loses electrons, and a nonmetal gains those electrons. And all of them form an anion with a single negative charge. The VIA elements gain two electrons to form anions with a 2- charge.
What is a anion example?
Anions are negatively charged ions. They are formed when non-metal gains the electrons. Therefore, they possess a net negative charge. Some examples of anions are Iodide (I–), chlorine (Cl–), hydroxide (OH–).
When anion is formed?
What is an anion? An anion has more electrons than protons, consequently giving it a net negative charge. For an anion to form, one or more electrons must be gained, typically pulled away from other atoms with a weaker affinity for them.
How do you identify an anion?
Tests for anions
- dissolve a small sample of the solid salt you are testing in water.
- place approximately 10cm 3 of the solution into a test tube.
- add four drops of nitric acid.
- add silver nitrate solution, dropwise.
- if a precipitate is produced, observe the colour.
How are cations created?
Cations form when an atom loses one or more electrons. The resulting cation has the electron configuration of the noble gas atom in the row above it in the periodic table.
When anions are formed their size does this?
The size of an anion will be larger than that of the parent atom because the addition of one or more electrons would result in increased repulsion among the electrons and decrease in effective nuclear charge. For example the ionic radius of F-is larger than F .
What is a basic anion?
• Basic anions – act as weak bases in water. – The anions (conjugate bases) of weak acids (F-, CN-, S2-, PO4. 3- …) → act as weak bases.
Do anion emitters work?
Although some research supports some positive effects of exposure to negative ions, there is no evidence-based medicine that supports negative ion therapy. So don’t bother getting any home negative ionizers. They can produce dangerous indoor ozone and just waste space and electricity.
How does an anion form in an atom?
An anion forms when it accepts an electron from another atom. Electrons are negatively charged, so the addition of an extra electron makes it so the… See full answer below. Become a Study.com member to unlock this answer! Create your account Already a member? Log in here
How are anions formed in the seventh period?
By contrast hydrogen, fluorine and iodine only need to gain one electron so they form an anion with a charge of negative one. Elements in the seventh period often form ionic bonds with metals, meaning that they take the electron from the metal and leave the metal with a positive charge.
How are names and formulas help anion names?
Anion Names and Formulas Help Anion Names and Formulas Metallic atoms hold some of their electrons relatively loosely, and as a result, they tend to lose electrons and form cations. In contrast, nonmetallic atoms attract electrons more strongly than metallic atoms, and so nonmetals tend to gain electrons and form anions.
What kind of ions can an anion gain?
If an anion has a charge of -2 or -3, it can gain one or two H + ions and still retain a negative charge. For example, carbonate, CO 32-, can gain an H + ion to form HCO 3-, which is found in baking soda.
What is the difference between an anion and a cation?
The key difference between anion and cation is that anions are the negatively charged ions formed from neutral atoms whereas cations are positively charged ions formed from neutral atoms. Commonly, anions and cations are called ions.
Which elements commonly form anions?
Common anions include acetate, hydroxide, carbonate, chlorate, cyanide, fluoride, nitrate, phosphate, and oxide. Except for hydroxide and cyanide, all that end in –ide are monatomic. Group 7 atoms called halides , which include such elements as fluorine , chlorine, bromine and iodine, form anions with a charge of negative one.
What is difference between anions and cations?
Difference Between Cation and Anion Definition. An anion is a negatively charged ion resulted by the acceptance of one or more electrons to its shells in the attempt to increase stability. Charge. Cations are positively charged. Form of Atom. Cations are usually formed by metal atoms. Electrolysis. Formation of Compounds.
How do you name anions?
The anion is named by taking the elemental name, removing the ending, and adding “ide.” For example, F -1 is called fluoride, for the elemental name, fluorine . The “ine” was removed and replaced with “ide.” To name a compound, the cation name and the anion named are added together.