How can isoenzymes be used clinically
Serum enzymes and isoenzymes are of clinical interest because they can be used as molecular markers of tissue damage. … Tissue damage which renders the cell membrane permeable will result in the leakage of soluble cell contents, including enzymes, into the serum.
What are isoenzymes and explain about their clinical significance?
Isoenzymes, or isozymes, are distinct, often readily separable forms of an enzyme elaborated by the same organism. Isozymes catalyze the same chemical reaction, but typically differ with respect to their primary structure, intracellular location, and physiological role.
What are isoenzymes in clinical biochemistry?
Isoenzymes (or isozymes) are a group of enzymes that catalyze the same reaction but have different enzyme forms and catalytic efficiencies.
What is the role of isoenzymes in diagnosis?
Because the enzyme composition of single organs is qualitatively and, to a certain extent, quantitatively similar, the diagnostic value of enzyme activity determinations is often diminished. Each serum enzyme can be separated into isoenzymes which stem from different organs and make specific organ diagnoses possible.What is the importance of isoenzymes?
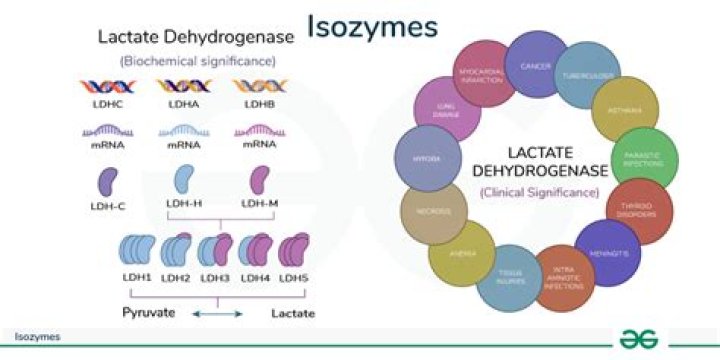
The existence of isozymes permits the fine-tuning of metabolism to meet the particular needs of a given tissue or developmental stage (for example lactate dehydrogenase (LDH)). In biochemistry, isozymes (or isoenzymes) are isoforms (closely related variants) of enzymes.
Which of the following property is not shown by isoenzyme?
4. Which of the following property is not shown by isoenzyme? Explanation: Sigmoidal shaped curve is observed when a plot of rate of reaction versus substrate concentration. This is as a property of allosteric enzyme.
What are the different isoenzymes?
- LDH-1: found in heart and red blood cells.
- LDH-2: found in white blood cells. …
- LDH-3: found in lung tissue.
- LDH-4: found in white blood cells, kidney and pancreas cells, and lymph nodes.
- LDH-5: found in the liver and muscles of skeleton.
How do you find isoenzymes?
These were originally developed for the cytochemical localization of enzymes (reviewed by Pearse, 1960), but have since found many applications for the detection of isoenzymes separated by micro-electrophoresis on starch or agar gel or on cellulose acetate.How do you identify isoenzymes?
To identify isozymes, a crude protein extract is made by grinding animal or plant tissue with an extraction buffer, and the components of extract are separated according to their charge by gel electrophoresis.
Do isoenzymes Localise in different tissues?Isoenzymes. Isoenzymes (also called isozymes) are alternative forms of the same enzyme activity that exist in different proportions in different tissues. Isoenzymes differ in amino acid composition and sequence and multimeric quaternary structure; mostly, but not always, they have similar (conserved) structures.
Article first time published onWhat are isozymes explain with example?
Isozymes (also known as isoenzymes) are homologous enzymes that catalyze the same reaction but differ in structure. … For example, the isoenzymes of lactate dehydrogenase in animal organs are different in term of their amino acid sequences and the level of their expression.
What are isoenzymes PPT?
ISOENZYMES Isoenzymes or isozymes are mutiple forms of same enzyme that catalyse the same chemical reaction Different chemical and physical properties: Electrophoretic mobility Kinetic properties Amino acid sequence Amino acid composition 2.
Which isoenzyme has fastest electrophoretic mobility?
mobility. The fastest anodally migrating isoenzyme is LD-1 (H4), and the slowest LD-5 (M4).
Which of the following is an example of isozyme?
α-amylase, glucokinase, lactate dehydrogenases all are the example of isozymes.
What are the advantages of utilizing isozymes in different tissues?
The advantages of isoenzymes is that it can catalyze the same reaction under the different environments within the different organelles. Isozymes are an important entity in metabolism for servicing a specific tissue or developmental sequence.
Which type of marker is considered in respect of isozyme?
Isozymes are protein markers. The technique is based on the principal that allelic variation exists from many different proteins.
Which LDH isoenzyme is elevated in myocardial infarction?
LDH-5: Highest amounts found in liver and skeletal muscle. Usually LDH isoenzyme levels increase 24–72 hours following myocardial infarction and reach a peak concentration in 3–4 days. The levels remain elevated for 8 to 14 days, making it a late marker for myocardial infarction.
What is difference between isoforms and isoenzymes?
Isoforms are highly related gene products that perform essentially the same biological function. Isozymes are isoforms of an enzyme. … Isoforms are almost always either the products of one gene or of multiple genes that evolved from a single ancestor gene.
Do isoenzymes have the same molecular weight?
Yes, this is very possible – isozymes of the same protein can have the same apparent MW on GF / SDS-PAGE. For example, they can differ at one amino acid position not affecting activity or (approximate) MW but, if the change involves a change in charge, it will be affecting pI.
Which of the following is an example of group transferring coenzyme?
An example of hydrogen transferring coenzyme is NAD+.
How are isoenzymes separated?
The separation of isozymes on the basis of surface charge (and to a lesser extent on molecular weight) may be achieved by electrophoresis in starch gel, acrylamide gel, agarose, cellulose acetate or Cellogel under conditions of pH, ionic strength, and ionic composition appropriate for a specific enzyme.
What are the 5 LDH isoenzymes?
There are five different forms of LDH that are called isoenzymes. They are distinguished by slight differences in their structure. The isoenzymes of LDH are LDH-1, LDH-2, LDH-3, LDH-4, and LDH-5. Different LDH isoenzymes are found in different body tissues.
What are creatine kinase isoenzymes?
Creatine kinase (CK) is an enzyme found in your muscles. … The 3 types of CK are called isoenzymes. They are: CK-MM, found in your skeletal muscle and heart. CK-MB, found in the heart and rises when heart muscle is damaged.
What does it mean if LDH is low?
Sometimes, LDH is used to monitor a cancer treatment and if LDH values decrease, it is a positive sign of the treatment. A low LDH blood level means: Moderate LDH decrease (< 130 UI/l in adults): LDH level is below the normal range and it may be due to an excessive vitamin C intake or due to a drug interference.
What is an isoenzyme pathway?
Isozymes or Isoenzymes are proteins with different structure which catalyze. the same reaction. Frequently they are oligomers made with different. polypeptide chains, so they usually differ in regulatory mechanisms and in. kinetic characteristics.
What are the characteristics of isoenzymes?
1. They catalyze the same reaction but they can be distinguished by physical methods such as electrophoresis or by immunological methods. 2. The difference between some isozymes are due to differences in the quarternary structure of the enzymes, e.g., lactate dehydrogenase exists in five isozymic forms.
How are isozymes regulated?
Isozymes arise from different genes, have different sequences of amino acids and a different structure yet catalyze the same reaction, have different properties and exhibit different enzymes kinetics and are usually controlled by different allosteric effectors.
Do substrates have an active site?
The part of the enzyme where the substrate binds is called the active site (since that’s where the catalytic “action” happens). A substrate enters the active site of the enzyme. This forms the enzyme-substrate complex.
Do isozymes share the same substrate?
As Dominique says, isoenzymes are defined in the first instance by the fact that they catalyse the same reaction, and they are likely to have different kinetic properties for this shared substrate, sometimes very different – e.g. hexokinase and glucokinase for glucose.
What are the enzymes of clinical significance?
Overall, LD, ALT, AST, ALP, and GGT are enzymes that are clinically significant in various disorders including conditions of the liver.
Who first coined the term enzyme?
2 Early enzymology-demystifying life. In 1833, diastase (a mixture of amylases) was the first enzyme to be discovered,2 quickly followed by other hydrolytic enzymes such as pepsin and invertase,3 but the term enzyme was only coined in 1877 by Wilhelm Kühne.