How do you draw the element aluminum?
How do you draw the element aluminum?
Starts here2:37How to Draw the Bohr-Rutherford Diagram of Aluminum – YouTubeYouTubeStart of suggested clipEnd of suggested clip56 second suggested clipAll right get your crayons out we’re gonna draw the border for diagram for aluminum aluminum on myMoreAll right get your crayons out we’re gonna draw the border for diagram for aluminum aluminum on my periodic table and hopefully yours. Too as an atomic number of 13.
What is aluminum Bohr model?
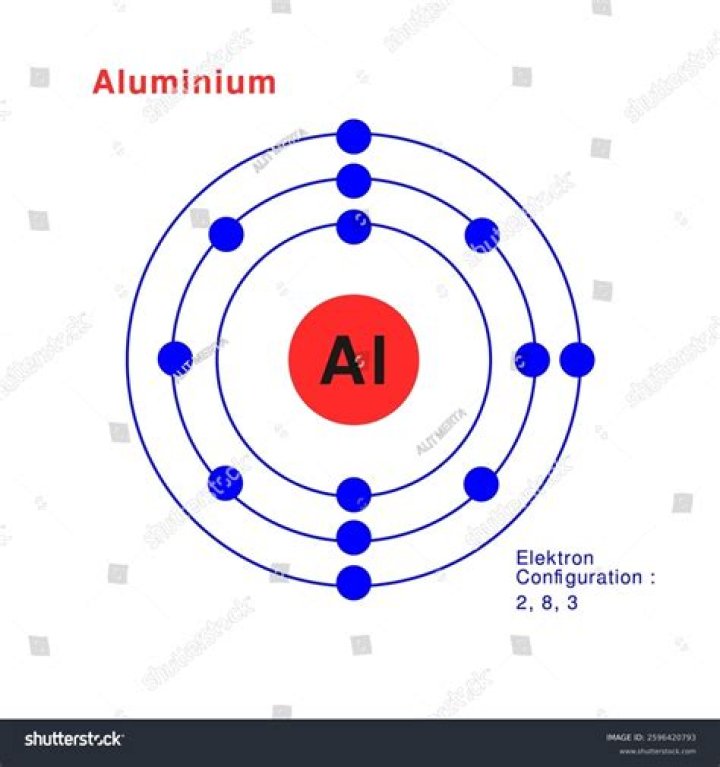
The Bohr model for aluminum shows its 13 protons and neutrons inside the nucleus with 13 electrons orbiting in 3 energy levels.
What are the elements of aluminum?
aluminum (Al), also spelled aluminium, chemical element, a lightweight silvery white metal of main Group 13 (IIIa, or boron group) of the periodic table….aluminum.
| atomic number | 13 |
|---|---|
| atomic weight | 26.9815384 |
| melting point | 660 °C (1,220 °F) |
| boiling point | 2,467 °C (4,473 °F) |
| specific gravity | 2.70 (at 20 °C [68 °F]) |
How do circles model an aluminum atom?
Aluminum’s atomic number is 13, thus each aluminum atom has 13 +(protons) and 13- (electrons). s the 13 electrons outside and at different distances from the nucleus. The red circle represent the nucleus and the black circles represent the energy levels away from the nucleus.
What is the electron number of Aluminium?
13 electrons
An aluminium atom has 13 electrons, arranged in an electron configuration of [Ne] 3s2 3p1, with three electrons beyond a stable noble gas configuration.
What does al3+ mean?
Aluminium
Description. Aluminium(3+) is an aluminium cation that has a charge of +3. It is an aluminium cation, a monoatomic trication and a monoatomic aluminium.
What element does the Bohr model represent?
The Bohr model of the hydrogen atom explains the connection between the quantization of photons and the quantized emission from atoms. Bohr described the hydrogen atom in terms of an electron moving in a circular orbit about a nucleus.
How does Bohr model work?
Summary. The Bohr model postulates that electrons orbit the nucleus at fixed energy levels. Orbits further from the nucleus exist at higher energy levels. When electrons return to a lower energy level, they emit energy in the form of light.
Is aluminum an element or compound or mixture?
Aluminum is a chemical element with symbol Al and atomic number 13. Classified as a post-transition metal, Aluminum is a solid at room temperature.
What is the most common isotope of aluminum?
The stable isotope of aluminum is aluminum-27. As all aluminum atoms have 13 protons in the nucleus, aluminum-27 must then have 14 neutrons. The radioactive isotope of aluminum that is also very common is aluminum-26, which has 13 neutrons.
Does helium have 2 neutrons?
Helium is the second element of the periodic table and thus is an atom with two protons in the nucleus. Most Helium atoms have two neutrons in addition to the protons. In its neutral state, Helium has two electrons in orbit about the nucleus.
What is the AR of Aluminium?
13
Aluminium (aluminum in American and Canadian English) is a chemical element with the symbol Al and atomic number 13….
| Aluminium | |
|---|---|
| Appearance | silvery gray metallic |
| Standard atomic weight Ar, std(Al) | 26.9815384(3) |
| Aluminium in the periodic table |