How do you find the heat of an exothermic reaction?
How do you find the heat of an exothermic reaction?
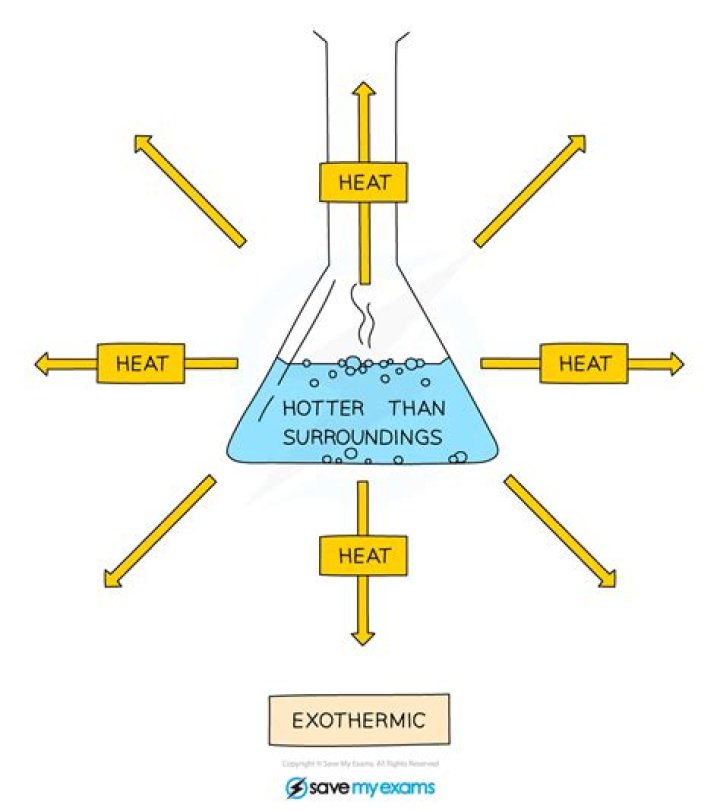
If the enthalpy change listed for a reaction is negative, then that reaction releases heat as it proceeds — the reaction is exothermic (exo- = out). If the enthalpy change listed for the reaction is positive, then that reaction absorbs heat as it proceeds — the reaction is endothermic (endo- = in).
How is exothermic measured?
Measurement. The enthalpy of a chemical system is essentially its energy. The heat release and corresponding energy change, Δ H , of a combustion reaction can be measured particularly accurately. The measured heat energy released in an exothermic reaction is converted to ΔH⚬ in Joule per mole (formerly cal/mol).
How is the heat of reaction measured?
The heat of reaction can be calculated based on the standard heat of formation of all reactants involved. However, it is usually determined by measuring the heat production over time using a reaction calorimeter, such as a heat flow calorimeter.
How do you know if heat is exothermic or endothermic?
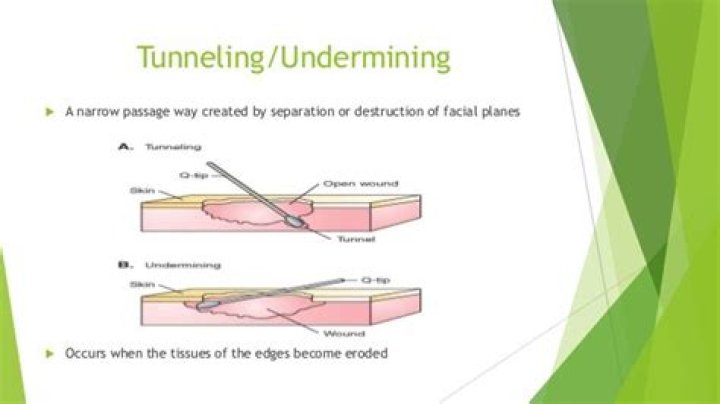
In a chemical equation, the location of the word “heat” can be used to quickly determine whether the reaction is endothermic or exothermic. If heat is released as a product of the reaction, the reaction is exothermic. If heat is listed on the side of the reactants, the reaction is endothermic.
Is exothermic hot or cold?
An exothermic process releases heat, causing the temperature of the immediate surroundings to rise. An endothermic process absorbs heat and cools the surroundings.”
Is Melting Chocolate endothermic?
For the first question, the correct answer among the choices listed is option A, a chocolate bar melting in hot car. It is an endothermic process because it absorbs heat energy for it to be melted.
Which is the exothermic process?
Exothermic reactions are reactions or processes that release energy, usually in the form of heat or light. In an exothermic reaction, energy is released because the total energy of the products is less than the total energy of the reactants.
Is heat of reaction positive or negative?
The heat of reaction is positive for an endothermic reaction.
How do I calculate heat?
Subtract the final and initial temperature to get the change in temperature (ΔT). Multiply the change in temperature with the mass of the sample. Divide the heat supplied/energy with the product. The formula is C = Q / (ΔT ⨉ m) .
Does exothermic get hot?
What is the hottest exothermic reaction?
The combustion of cyanogen 4795 °K or dicyanoacetylene 5260 °K in pure oxygen produces extremely hot flames.
How is heat flow measured in an exothermic reaction?
(B) In an exothermic reaction, heat flows from the system into the surroundings, increasing the temperature of the surroundings. Heat flow is measured in one of two common units: the calorie and the joule. The joule (J) is the SI unit of energy.
How is the heat flow in a calorimeter calculated?
Ti < Tf, reaction gives heat to water and make increase its temperature. Thus, it is exothermic reaction. Ti > Tf, reaction absorbs heat from water and make decrease its temperature. Thus, it is endothermic reaction. Heat flow in calorimeter is calculated with following formula; Q=mcal.ccal.∆T + mwater. cwater.∆T Where;
How is the heat of a chemical reaction measured?
Heat of Reaction. The Heat of Reaction (also known and Enthalpy of Reaction) is the change in the enthalpy of a chemical reaction that occurs at a constant pressure. It is a thermodynamic unit of measurement useful for calculating the amount of energy per mole either released or produced in a reaction.
How is energy transferred in an exothermic reaction?
An exothermic reaction is a reaction in which energy is released in the form of light or heat. Thus in an exothermic reaction, energy is transferred into the surroundings rather than taking energy from the surroundings as in an endothermic reaction. In an exothermic reaction, change in enthalpy (ΔH) will be negative.
(B) In an exothermic reaction, heat flows from the system into the surroundings, increasing the temperature of the surroundings. Heat flow is measured in one of two common units: the calorie and the joule. The joule (J) is the SI unit of energy.
Ti < Tf, reaction gives heat to water and make increase its temperature. Thus, it is exothermic reaction. Ti > Tf, reaction absorbs heat from water and make decrease its temperature. Thus, it is endothermic reaction. Heat flow in calorimeter is calculated with following formula; Q=mcal.ccal.∆T + mwater. cwater.∆T Where;
An exothermic reaction is a reaction in which energy is released in the form of light or heat. Thus in an exothermic reaction, energy is transferred into the surroundings rather than taking energy from the surroundings as in an endothermic reaction. In an exothermic reaction, change in enthalpy (ΔH) will be negative.
How is the net amount of heat released in a chemical reaction measured?
When a calorimeter, a device used to measure the heat released by a chemical reaction, the net amount of heat energy that flows through the device is equal to the negative of the total energy change of the system. However, it is extremely difficult to measure or even calculate the absolute total of energy in a given chemical system.