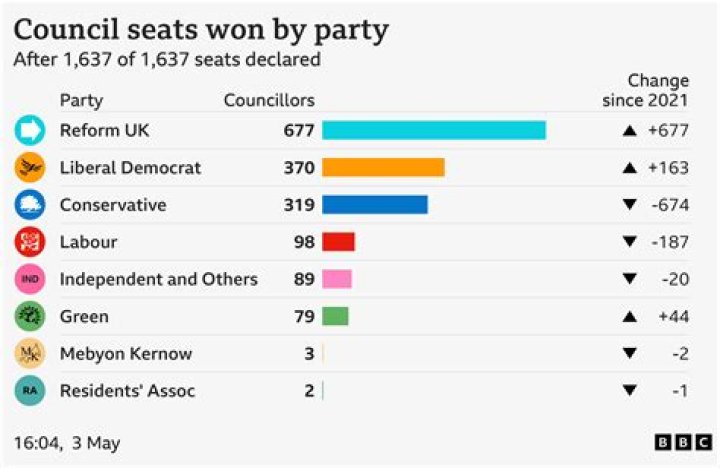
How do you identify a dative covalent bond?

How do you identify a dative covalent bond?
A dative bond is also known as a dipolar bond or coordinate bond. In a diagram, a dative bond is indicated by drawing an arrow pointing from the atom that donates the lone electron pair toward the atom that accepts the pair. The arrow replaces the usual line that indicates a chemical bond.
Which compound contains a coordinate dative covalent bond?
ammonium ion
It is also known as dative bonding and it is a directional bond i.e., sharing of electrons takes place in a definite direction. Hence, the compound which contains covalent coordinate bonds is ammonium ion.
What are some examples of coordinate covalent bonds?
Here are a few examples of the coordinate covalent bond.
- Formation Of Ammonium Ion. The nitrogen atom in Ammonia donates its electron pair to the empty orbital of H+ ion thus nitrogen is donor, H+ is acceptor and a co-ordinate bond is formed.
- Formation Of Hydronium Ion.
- Formation Of Ammonia Boron Trifluoride.
What is meant by dative covalent bond?
A coordinate covalent bond, also known as a dative bond, dipolar bond, or coordinate bond is a kind of two-center, two-electron covalent bond in which the two electrons derive from the same atom. The bonding of metal ions to ligands involves this kind of interaction.
How is dative covalent bond formed?
A dative covalent is formed when the pair of electrons forming the bond are donated by one atom only. This contrasts with the usual covalent bond by each atom of the bond contributing one electron.
Which species have a dative covalent bond?
An example of a dative covalent bond is provided by the interaction between a molecule of ammonia, a Lewis base with a lone pair of electrons on the nitrogen atom, and boron trifluoride, a Lewis acid by virtue of the boron atom having an incomplete octet of electrons.
Why is there no dative bond in co3 2?
Carbonato cannot form a coordinate covalent bond using the O atom in the double C-O bond because oxygen is most stable when it has two bonding pairs and two non-bonding pairs. That O atom is already in a favorable state and would not likely want to form a coordinate bond.
What are 5 Covalent compounds?
Binary molecular (covalent) compounds
| Prefixes used in chemical nomenclature | |
|---|---|
| prefix | number of atoms |
| penta- | 5 |
| hexa- | 6 |
| hepta- | 7 |
Is dative bond a covalent bond?
How do dative covalent bonds form?
A coordinate bond (also called a dative covalent bond) is a covalent bond (a shared pair of electrons) in which both electrons come from the same atom. A covalent bond is formed by two atoms sharing a pair of electrons. The atoms are held together because the electron pair is attracted by both of the nuclei.
What are the properties of dative bond?
(1) Their melting and boiling points are higher than purely covalent compounds and lower than purely ionic compounds. (2) These are sparingly soluble in polar solvent like water but readily soluble in non-polar solvents. (3) Like covalent compounds, these are also bad conductors of electricity.