How does ionizing radiation create ions
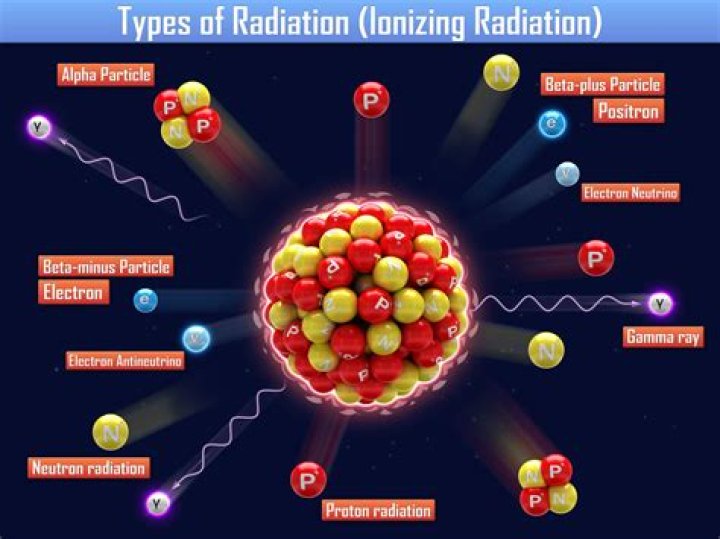
If an electron is “knocked” out of its orbit it leaves a positively charged ion. When ionizing radiation passes through anything, it can knock electrons out of their orbits, forming ions; hence its name. Some energy from the ionizing radiation is used up each time an electron is knocked from its orbit.
How are ions produced by ionizing radiation?
When ionizing radiation passes through material such as air, water, or living tissue, it deposits enough energy to produce ions by breaking molecular bonds and displace (or remove) electrons from atoms or molecules. … This electron displacement may lead to changes in living cells.
How does ionizing radiation affect an atom?
Ionizing radiation describes atoms becoming ionized to ions. During ionizing radiation, an electron is ejected off the atom, causing the atom to lose an electron and become ionized. Non-ionizing radiation is generally caused by excitation of electrons.
What does ionizing radiation create?
Exposure to ionizing radiation causes cell damage to living tissue. In high acute doses, it will result in radiation burns and radiation sickness, and lower level doses over a protracted time can cause cancer.What ions are in radiation?
Cosmic radiation that penetrates the Earth’s atmosphere from space consists mainly of protons, alpha particles, and heavier atomic nuclei. Positrons, mesons, pions, and other exotic particles can also be ionizing radiation.
Why is ionizing radiation called ionizing?
A familiar example of ionizing radiation is that of x-rays, which can penetrate our body and reveal pictures of our bones. We say that x-rays are “ionizing,” meaning that they have the unique capability to remove electrons from atoms and molecules in the matter through which they pass.
At what frequency does radiation become ionizing?
UV radiation above 10 eV (wavelength shorter than 125 nm) is considered ionizing. However, the rest of the UV spectrum from 3.1 eV (400 nm) to 10 eV, although technically non-ionizing, can produce photochemical reactions that are damaging to molecules by means other than simple heat.
What does it mean if an atom is ionized?
Atoms are comprised of a nucleus of protons and neutrons, which can be thought of as surrounded by a cloud of orbiting electrons. When one (or more) electron is stripped off or added to the atom, it is no longer electrically neutral and an ion is formed; the atom is said to be ionised.Why is it called ionizing radiation?
In this context, an unstable atom is said to be “radioactive,” and the energy it releases is referred to as “radiation.” When the radiation has enough energy to ionize other atoms (i.e., remove negatively-charged particles called “electrons”) in its path, it is referred to as “ionizing radiation.” Types of ionizing …
What is the importance of ionizing and nonionizing radiation?Non-ionizing radiation is longer wavelength/lower frequency lower energy. … Ionizing Radiation has sufficient energy to produce ions in matter at the molecular level. If that matter is a human significant damage can result including damage to DNA and denaturation of proteins.
Article first time published onWhat collects on the ions in a cloud chamber?
How does radiation produce ions in matter? … What collects on the ions in a cloud chamber? Droplets of vapor. What is emitted when a material scintillates?
What radioactive particles are primarily responsible for causing ionization?
Radiation that has enough energy to remove or knock out electrons from atoms, and thus create positively charged ions is known as ionizing radiation. Alpha and beta particles are the radioactive particles that cause ionizing radiation to occur.
Can electrons cause ionization?
The energetic electrons resulting from the absorption of radiant energy and the passage of charged particles in turn may cause further ionization, called secondary ionization.
What are the three ionizing radiation?
There are three main kinds of ionising radiation: alpha particles, which include two protons and two neutrons. beta particles, which are essentially electrons. gamma rays and x-rays, which are pure energy (photons).
What is the difference between ionizing and non ionizing radiation *?
The main difference between ionizing and nonionizing radiation is that ionizing radiation refers to types of radiation where the radiation carries enough energy to ionize atoms, whereas nonionizing radiation refers to types of radiation that do not carry enough energy to ionize atoms.
What is the strongest ionizing radiation?
Alpha particles have approximately four times the mass of a proton or neutron and approximately ~8,000 times the mass of a beta particle (Figure 5.4. 1). Because of the large mass of the alpha particle, it has the highest ionizing power and the greatest ability to damage tissue.
Is UV Ionising?
Ultraviolet (UV) radiation is a form of non-ionizing radiation that is emitted by the sun and artificial sources, such as tanning beds. While it has some benefits for people, including the creation of Vitamin D, it also can cause health risks.
Do cell phones emit ionizing radiation?
Cell phones emit low levels of radio frequency energy, a type of non-ionizing radiation. The available scientific data on exposure to radio frequency energy show no categorical proof of any adverse biological effects other than tissue heating.
What are the benefits of ionizing radiation?
Ionizing radiation has so much energy it can knock electrons out of atoms. But, when used properly, ionizing radiation has useful applications in the world around us. Food irradiation does not make food radioactive, but kills harmful bacteria that can make us sick. Nuclear medicine can help treat illnesses like cancer.
Which is true about ionizing radiations?
All of the above are true. Ionizing radiation is energy that is released by an unstable atom in order to become more stable. … Ionizing radiation has enough energy to remove electrons from atoms in its path.
What is Ionising power in physics?
Ionising power refers to how well can it knock out electrons from target nuclei. Penetration power is how far can the ray travel before losing all energy. \alpha particles can be stopped by the human skin. \beta particcles can be stopped by a aluminium foil. \gamma rays can be stopped by a thick block of Pb.
What type of radiation is ionizing?
Ionizing radiation takes a few forms: Alpha, beta, and neutron particles, and gamma and X-rays. All types are caused by unstable atoms, which have either an excess of energy or mass (or both). In order to reach a stable state, they must release that extra energy or mass in the form of radiation.
How atoms are converted into ions?
An atom becomes an Ion (a) if it gains one or more electron(s) or (b) if it loses one or more electron(s). When it gains electrons it becomes negatively charged and is called an anion. When it loses electron(s) it becomes positively charged and is called a cation.
How are ions formed in the environment?
Formation of monatomic ions Monatomic ions are formed by the gain or loss of electrons to the valence shell (the outer-most electron shell) in an atom.
What is the difference between oxidation and ionization?
Ionization energy is the amount of energy required to remove an electron. Oxidation potential is the opposite of the reduction potential, which is electrical potential (i.e., voltage) derived from comparing the spontaneity of the reduction compared to reducing a standard hydrogen electrode.
Does ionizing radiation produce heat?
Biological Effects of Ionizing Radiation Radiation in or near the visible portion of the spectrum excites electrons into higher-energy orbitals. When the electron eventually falls back to a lower-energy state, the excess energy is given off to neighboring molecules in the form of heat.
Why ionizing radiation does more harm than non-ionizing radiation?
Non-ionizing radiation has enough energy to move atoms in a molecule around or cause them to vibrate, but not enough to remove electrons from atoms. … Ionizing radiation can affect the atoms in living things, so it poses a health risk by damaging tissue and DNA in genes.
Why ionizing radiation is more effective over the non-ionizing radiation in case of sterilization?
Ionizing radiation is the use of short wavelength, high-intensity radiation to destroy microorganisms. … Non-ionizing radiation uses longer wavelength and lower energy. As a result, non-ionizing radiation loses the ability to penetrate substances, and can only be used for sterilizing surfaces.
How does an ion chamber work?
The operating principle of an ionization chamber is simple: ionizing radiation from the source (X- or gamma rays, electrons) creates an ionization of the gas atoms. A voltage is applied between the electrodes. Negative charges are attracted by the anode, positive charges by the cathode.
What are ionizing particles?
RADIATION QUANTITIES AND UNITS Direct ionizing particles are charged particles, such as electrons, protons, and alpha particles, having sufficient kinetic energy to produce ionization by collision.
What is the purpose of making cloud chamber?
Cloud chambers are used to detect ionising particles. These can include any electrically charged particle that passes through the chamber; and the amount of ionisation can be deduced from the tracks in the chamber and used to determine a particle’s properties and identity.