How does osmotic pressure relate to Osmosis
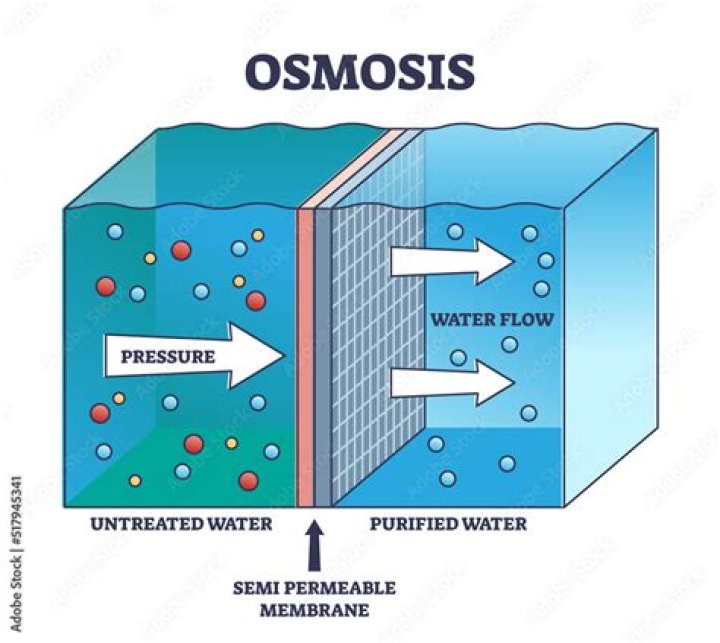
Osmosis is defined as the net flow or movement of solvent molecules through a semipermeable membrane through which solute molecules cannot pass. … Osmotic pressure is the pressure that needs to be applied to a solution to prevent the inward flow of water across a semipermeable membrane.
How osmotic pressure is related to?
Osmotic pressure is the minimum pressure which needs to be applied to a solution to prevent the inward flow of its pure solvent across a semipermeable membrane. It is also defined as the measure of the tendency of a solution to take in a pure solvent by osmosis.
Is osmotic pressure the driving force of osmosis?
The physical driving force of osmosis is the increase in entropy generated by the movement of free water molecules. There is also thought that the interaction of solute particles with membrane pores is involved in generating a negative pressure, which is the osmotic pressure driving the flow of water.
What happens if osmotic pressure is high?
loss of electrolytes (salt), the osmotic pressure of the extracellular fluids becomes higher than in the cells. Since water passes from a region of lower to a region of higher osmotic pressure, water flows out of the cells into the extracellular fluid, tending to lower its osmotic pressure and increase…Is osmotic pressure and osmotic potential same?
Osmotic Pressure (OP): The actual pressure, that develops in a solution, when it is separated from pure water by means of semipermeable membrane. … Osmotic potential: It is the decrease in chemical potential of pure water due to the presence of solute particle in it.
How does osmotic pressure affect blood pressure?
When your body senses either an increase in osmolarity, a decrease in blood pressure, or both, it reacts with different homeostatic mechanisms to try to increase water volume back to normal levels, restore blood pressure, and ensure adequate circulation.
Is osmotic pressure the same as water potential?
The key difference between water potential and osmotic potential is that water potential is the measure of the concentration of free water molecules while the osmotic potential is the measure of the tendency of a solution to withdraw water from pure water through a semi-permeable membrane via osmosis.
What causes osmosis?
Osmosis occurs according to the concentration gradient of water across the membrane, which is inversely proportional to the concentration of solutes. … Osmosis occurs when there is a concentration gradient of a solute within a solution, but the membrane does not allow diffusion of the solute.What does osmotic pressure mean?
Osmotic pressure is defined as the pressure that must be applied to the solution side to stop fluid movement when a semipermeable membrane separates a solution from pure water.
Does osmotic pressure move water?Water will move along its concentration gradient. This force is termed osmotic pressure or, in the case of colloids, e.g., albumin, oncotic pressure. It is proportional to the number of atoms/ions/molecules in solution and is expressed as mOsm/liter (osmolarity) or mOsm/kg (osmolality) of solution.
Article first time published onWhat is the driving force of osmosis called?
The driving force for this separation is an osmotic pressure gradient, such that a “draw” solution of high concentration (relative to that of the feed solution), is used to induce a net flow of water through the membrane into the draw solution, thus effectively separating the feed water from its solutes.
Why osmotic pressure is positive and osmotic potential is negative?
Osmotic pressure is a hydrostatic pressure exerted to the solution to prevent the flow of water through the semi permeable membrane. … As a solution will always have some solute, the amout of water will be lesser than pure water. Thus the osmotic potential of a solution will always be negative (value less than zero).
How is osmosis pressure different from osmotic potential?
Osmotic PressureOsmotic PotentialDevelops in a closed systemDevelops in closed or open systemValue is positiveValue is negative
What is the difference between osmosis and Exosmosis?
EndosmosisExosmosisThe direction of solvent: From the surrounding solution into the cell.The direction of solvent: From the cell into the surrounding solution.
What is the difference between osmotic pressure and turgor pressure?
Note: Osmotic pressure refers to the minimum pressure that needs to be applied on the solution to prevent the inward flow of its pure solvent across a semipermeable membrane whereas turgor pressure refers to the pressure inside the cell pushes the plasma membrane against the cell wall of the plant cell.
How does water potential relate to osmosis?
The water always moves from the region of higher water potential to the region of lower water potential. So the rate of osmosis is directly proportional to the water potential. If a solution has high water potential (low solute concentration) then osmosis will take place.
What is osmosis in terms of water potential?
Water Potential measures the concentration of free water molecules. It is a measure of the tendency of these molecules to diffuse to another area. … Osmosis can therefore be defined as the diffusion of water from a region of high Water Potential to a region of low Water Potential through a Partially Permeable Membrane.
How does pressure affect water potential?
When the amount of solutes increases, osmotic potential decreases, and total water potential decreases. When the pressure increases, water potential increases. Both of these can be used to decrease water potential in specific areas, forcing the movement of high potential water into various cells of the plants.
How does osmotic pressure relate to Oedema?
Increased matrix density also increases the excluded volume, which acts to increase the effective interstitial colloid osmotic pressure. In effect, these changes create a suction force that accelerates fluid filtration and the development of edema.
What do you mean by osmosis osmotic pressure and isotonic solution?
An isotonic solution is one that has the same concentration of solutes both inside and outside the cell. A hypertonic solution is one that has a higher solute concentration outside the cell than inside. A hypotonic solution is the one that has a higher solute concentration inside the cell than outside.
How does albumin regulate osmotic pressure?
serum albumin, protein found in blood plasma that helps maintain the osmotic pressure between the blood vessels and tissues. … Circulating blood tends to force fluid out of the blood vessels and into the tissues, where it results in edema (swelling from excess fluid).
How does osmotic pressure depends on temperature and atmospheric pressure?
Osmotic pressure is driven by the tendency of the solute molecules that are not glueing together to take up as much space as they can get. … All else staying equal, this gives a direct proportional relationship between temperature and pressure.
What is osmotic pressure and give its relation with concentration of solution?
The osmotic pressure of a solution is the pressure difference needed to stop the flow of solvent across a semipermeable membrane. The osmotic pressure of a solution is proportional to the molar concentration of the solute particles in solution.
What is osmotic pressure kid definition?
Osmotic pressure is defined as the external pressure required to be applied so that there is no net movement of solvent across the membrane. … Osmotic pressure is a colligative property, meaning that the osmotic pressure depends on the molar concentration of the solute but not on its identity.
What is tonicity and osmosis?
“Tonicity is the ability of a solution to affect the fluid volume and pressure in a cell. If a solute cannot pass through a plasma membrane, but remains more concentrated on one side of the membrane than on the other, it causes osmosis.”
What would happen without osmosis?
Without osmosis your cells would not be able to have the proper levels of water to work at their best. … Or could possibly lead to a very dangerous condition called hyponatremia , which can cause cells to take in too much water diluting important electrolytes like sodium.
How do you explain osmosis?
In biology, osmosis is the movement of water molecules from a solution with a high concentration of water molecules to a solution with a lower concentration of water molecules, through a cell’s partially permeable membrane.
Why does water move from low osmotic pressure to osmotic pressure?
Initially, the level is the same in both arms of the U tube. As osmosis starts, it pushes the solvent from right arm to left arm and pushes the solution up even though the membrane is permeable to solvent until the osmotic pressure in the right arm is equal to the hydrostatic pressure exerted by the left arm.
Why does water flow from low to high osmotic pressure?
The substance flowing is water. Osmotic pressure is the pressure required to stop the flow of water. The osmotic pressure is highest where the water concentration is lowest*. That’s why water moves toward higher areas of osmotic pressure—it’s doing what all substances “try” to do—flow from high to low concentration.
Why does osmotic pressure stay the same?
Osmotic pressure is the “pulling” force on water due to the presence of solutes in solution. … Osmotic pressure remains relatively constant over the length of the capillary, but hydrostatic pressure drops sharply as it nears the venule end due to the initial loss of fluid volume.
How is osmosis different from diffusion?
Osmosis only allows solvent molecules to move freely, but diffusion allows both solvent and solute molecules to move freely. … Osmosis happens when molecules move from higher to lower concentrations, but diffusion happens when it is reversed.