How does the collision theory affect the rate of reaction?
How does the collision theory affect the rate of reaction?
Collision theory states that the rate of a chemical reaction is proportional to the number of collisions between reactant molecules. The more often reactant molecules collide, the more often they react with one another, and the faster the reaction rate.
What is the collision theory GCSE?
Collision theory states that chemical reactions occur only when the reactant particles collide with sufficient energy to react. The minimum amount of energy needed is called the activation energy, which is different for each reaction.
What 4 factors affect the rate of a reaction?
The factors that affect reaction rates are:
- surface area of a solid reactant.
- concentration or pressure of a reactant.
- temperature.
- nature of the reactants.
- presence/absence of a catalyst.
When every collision between reactants leads to a reaction what determines the rate at which the reaction occurs?
The rate of the reaction depends on the frequency of collisions. The theory also tells us that reacting particles often collide without reacting. For collisions to be successful, reacting particles must (1) collide with (2) sufficient energy, and (3) with the proper orientation.
What factors affect the collision theory?
There are several factors that affect reaction rates. Their effects can be explained using collision theory. These factors are the nature of the reactants, concentration, surface area, temperature and catalysts. Each of these factors increases reaction rate because they increase the number or energy of collisions.
How do you determine the rate of a reaction?
Measuring Reaction Rates
- Reaction rate is calculated using the formula rate = Δ[C]/Δt, where Δ[C] is the change in product concentration during time period Δt.
- The rate of reaction can be observed by watching the disappearance of a reactant or the appearance of a product over time.
What are 5 factors that affect reaction rate?
Five factors typically affecting the rates of chemical reactions will be explored in this section: the chemical nature of the reacting substances, the state of subdivision (one large lump versus many small particles) of the reactants, the temperature of the reactants, the concentration of the reactants, and the …
What three factors affect the rate of reaction?
The rate of a chemical reaction is influenced by many different factors, including reactant concentration, surface area, temperature, and catalysts.
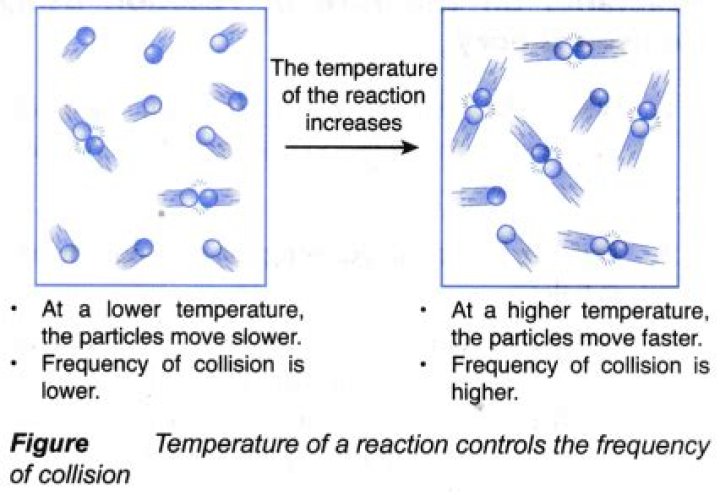
How is collision theory related to temperature and concentration of reactants?
The collision theory explains why reactions occur between atoms, ions, and molecules. With an increase in temperature, there is an increase in the number of collisions. Increasing the concentration of a reactant increases the frequency of collisions between reactants and will, therefore, increase the reaction rate.



