How many electrons are there in SPDF?
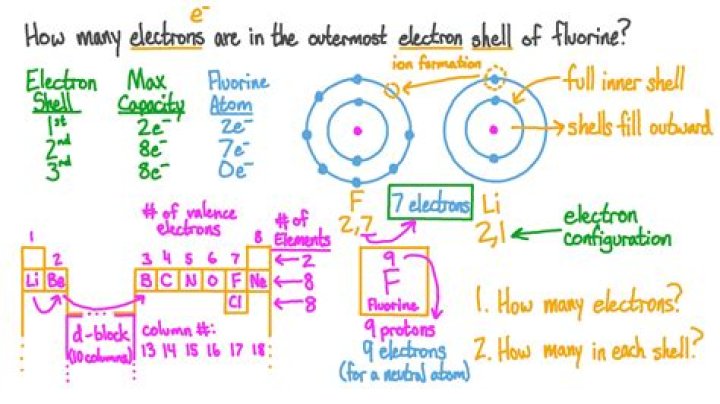
How many electrons are there in SPDF?
In this way, s subshell has two electrons, p subshell has six electrons, d subshell has ten electrons and f subshell has fourteen electrons in total.
What are SPDF electrons?
5 Answers. s, p, d, f and so on are the names given to the orbitals that hold the electrons in atoms. These orbitals have different shapes (e.g. electron density distributions in space) and energies (e.g. 1s is lower energy than 2s which is lower energy than 3s; 2s is lower energy than 2p).
What does 1s 2s 2p mean?
The superscript is the number of electrons in the level. The number in front of the energy level indicates relative energy. For example, 1s is lower energy than 2s, which in turn is lower energy than 2p. The number in front of the energy level also indicates its distance from the nucleus.
How many electrons can f carry?
14 electrons
An f subshell can hold up to 14 electrons.
What are the SPDF block elements?
S-block comprises 14 elements namely hydrogen (H), lithium (Li), helium (He), sodium (Na), beryllium (Be), potassium (K), magnesium (Mg), rubidium (Rb), calcium (Ca), cesium (Cs), strontium (Sr), francium (Fr), barium (Ba), and radium (Ra)….⇒ Check more:
- Actinides.
- Lanthanides.
- F Block Elements.
What is SPDF in periodic table?
From Wikipedia, the free encyclopedia. Spdf or SPDF may refer to: Electron configuration, for which there is an obsolete system of categorizing spectral lines as “sharp”, “principal”, “diffuse” and “fundamental”; also the names of the sub shells or orbitals. The blocks of the periodic table.
How do you write SPDF element?
spdf Notation To write the electron configuration of an atom, identify the energy level of interest and write the number of electrons in the energy level as its superscript as follows: 1s2. This is the electron configuration of helium; it denotes a full s orbital.
Can F hold 14 electrons?
The f sublevel as a whole can hold up to 14 electrons due to the fact that it consists of 7 orbitals, but each one can only hold up to 2 electrons.
What can hold up to 8 electrons?
Notice that lithium is the first element in the second row of the periodic table. The second shell has two subshells (labeled 2s and 2p). The 2s subshell holds a maximum of 2 electrons, and the 2p subshell holds a maximum of 6 electrons. This means that the second shell can hold a maximum of eight electrons (2+6=8).