How many isomers are possible for 1 Hexyne?
How many isomers are possible for 1 Hexyne?
All five hexane constitutional isomers have the same molecular formula, C6H14, and the same molecular weight, 86. However, each one of the hexanes has a unique boiling point….Constitutional (structural) Isomers:
| # of Carbons | Acyclic Alkane | # of Isomers |
|---|---|---|
| 3 | propane | 1 |
| 4 | butane | 2 |
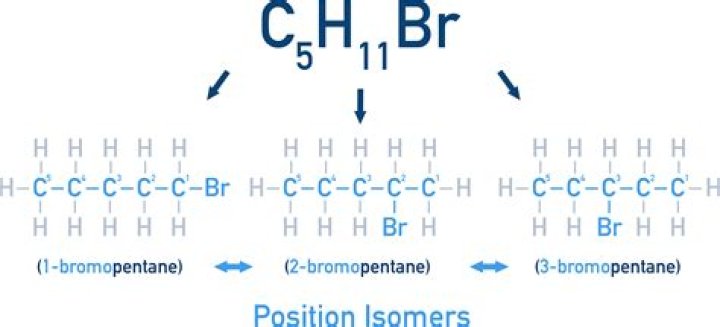
| 5 | pentane | 3 |
| 6 | hexane | 5 |
What type of isomers are 1-hexene?
One of the most common industrially useful isomers is 1-hexene, an alpha-olefin. Hexene is used as a comonomer in the production of polyethylene.
Can isomers be the same molecule?
Isomers are molecules that have the same molecular formula, but have a different arrangement of the atoms in space. If you can make an apparently different molecule just by rotating single bonds, it’s not different – it’s still the same molecule.
Does 1-hexene have geometric isomers?
1-hexene does not exhibit geometric isomerism. It cannot form cis-trans isomers because the first carbon carrying the double bond has no substituent attached to it.
What is the molecular formula of 1-Hexyne?
C6H10
1-Hexyne/Formula
What does 1-Hexyne look like?
1-Hexyne (n-butylacetylene) is a hydrocarbon consisting of a straight six-carbon chain having a terminal alkyne. Its molecular formula is C6H10. It is a liquid at room temperature that is colorless or pale yellow in appearance.
What is the density of 1-hexene?
673 kg/m³
1-Hexene/Density
What is the difference between stereoisomers and structural isomers?
Structural (constitutional) isomers have the same molecular formula but a different bonding arrangement among the atoms. Stereoisomers have identical molecular formulas and arrangements of atoms. They differ from each other only in the spatial orientation of groups in the molecule.
What is the molecular formula of 1-hexene?
C6H12
1-Hexene/Formula



