How many peaks are in the mass spectrum of cl2?
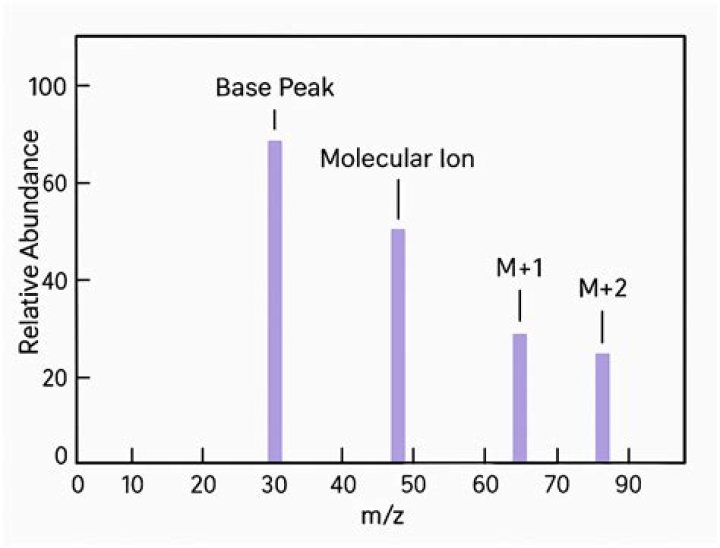
How many peaks are in the mass spectrum of cl2?
There are five main peaks of isotopes of chlorine of various isotopic monatomic ions. The presence of five peaks for chlorine shows the ratio of heights for peaks 1 and 2 is 3: 1. For bimolecular ions, ¾ of the chlorine isotopes are Cl 35 and ¼ of the isotope of chlorine is cl37.
What is the isotopic ratio of Cl in a mass spectrum?
3 : 1 ratio
The molar mass of chlorine is 35.4527 g/mol, but the mass spectrum shows us that a sample of chlorine atoms actually consists of 35Cl and 37Cl in a 3 : 1 ratio. What is the main difference between isotopes of an element?
What is the mass of chlorine?
35.453 u
Chlorine/Atomic mass
What is the mass of CL 35?
34.968852
Reference E95
| Isotope | Mass | Spin |
|---|---|---|
| 35Cl | 34.968852 | 3/2 |
| 37Cl | 36.965903 | 3/2 |
What mean MZ?
M stands for mass and Z stands for charge number of ions. In mass analysis, an electron is taken from molecules to create single charged ions. If two electrons are removed, double charged ions are produced. The number of electrons removed is the charge number (for positive ions).
What is the m 2 peak?
mass spectra – the M+2 peak. MASS SPECTRA – THE M+2 PEAK. This page explains how the M+2 peak in a mass spectrum arises from the presence of chlorine or bromine atoms in an organic compound. It also deals briefly with the origin of the M+4 peak in compounds containing two chlorine atoms.
How do you calculate isotopic abundance?
The equation can be set up as a percent or as a decimal. As a percent, the equation would be: (x) + (100-x) = 100, where the 100 designates the total percent in nature. If you set the equation as a decimal, this means the abundance would be equal to 1. The equation would then become: x + (1 – x) = 1.
How do you find the isotopic abundance of a mass spectrum?
The relative abundance of each isotope can be determined using mass spectrometry. A mass spectrometer ionizes atoms and molecules with a high-energy electron beam and then deflects the ions through a magnetic field based on their mass-to-charge ratios ( m / z m/z m/z ).
How many grams are in chlorine?
One mole of chlorine weighs 70 grams.
What is the mass of chlorine 37?
36.966 amu
Because they differ in the number of neutrons in the nucleus, isotopes of the same element differ in their atomic mass. For example, chlorine occurs naturally as a mixture of Cl-35, atomic mass = 34.969 amu (75.53%), and Cl-37, atomic mass = 36.966 amu (24.47%).