How many stereoisomers are possible for cyclohexane?
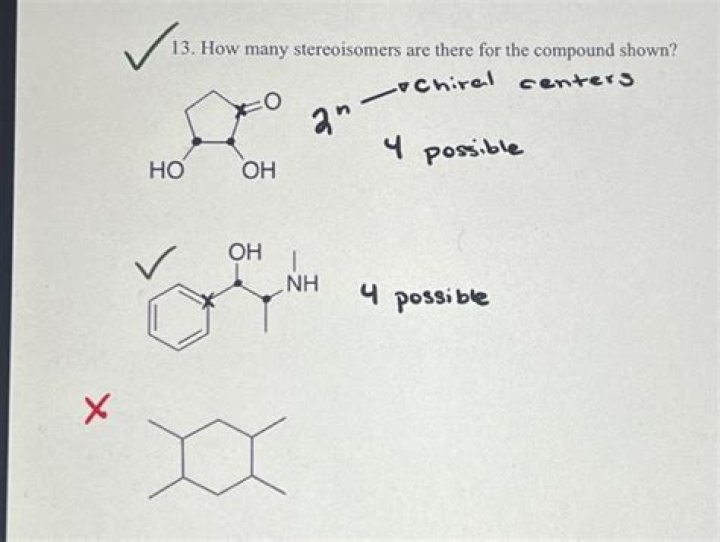
How many stereoisomers are possible for cyclohexane?
three
There are three configurational isomers of 1,2-dichlorocyclohexane and three configurational isomers of 1,3-dichlorocyclohexane. These are shown in the following table.
What is the fastest way to identify diastereomers?
Among molecules with the same connectivity:
- Molecules that are mirror images but non-superimposable are enantiomers.
- If they aren’t superimposable, and they aren’t mirror images, then they’re diastereomers.
How do you calculate diastereomers?
2n−2=24−2=16 – 2=14 (14 diastereomers). For example, D-glucose has 4 chiral carbons, so there are 16 aldohexoses (8 D and 8 L). L-Glucose is an enantiomer of D-glucose, and the other 14 aldohexoses are diastereomers of them.
What are diastereomers with examples?
Diastereomers may often include compounds which are ring structures. Imagine, for example, two compounds with a six-membered ring, each with two substituents, a chlorine atom and an ethyl group. They are also not mirror images of each other, just like our previous example, which defines them as diastereomers.
Can Cyclohexanes have diastereomers?
Diastereomer: One molecule in a set of stereoisomers that are not enantiomers. The cis and trans stereoisomers of 1,2-dichlorocyclohexane are diastereomers.
Can diastereomers be Achiral?
Achiral Diastereomers (meso-Compounds) Two of these stereoisomers are enantiomers and the third is an achiral diastereomer, called a meso compound. Meso compounds are achiral (optically inactive) diastereomers of chiral stereoisomers.
What are diastereomers pairs?
Diastereomers are defined as non-mirror image non-identical stereoisomers. Diastereomers differ from enantiomers in that the latter are pairs of stereoisomers that differ in all stereocenters and are therefore mirror images of one another.
How many diastereomers are possible for?
So, there are six diastereomers possible in the given structural molecule. According to the above calculation, we conclude there are six diastereomers possible in the given structural molecule. Hence, option C is the correct answer.
What are erythro and threo diastereomers?
Two older prefixes still commonly used to distinguish diastereomers are threo and erythro. In the case of saccharides, when drawn in the Fischer projection the erythro isomer has two identical substituents on the same side and the threo isomer has them on opposite sides.
Are E and Z diastereomers?
Cis/trans isomers or (E/Z) isomers are diastereomers, because they are stereoisomers of the same constitution, that is, the connections between every atom are the same, and the molecules do not have a mirror-image relationship.