How many valence electrons does magnesium have?
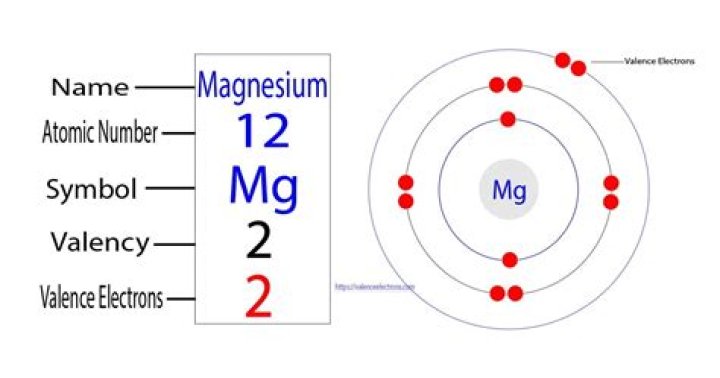
How many valence electrons does magnesium have?
Valence electrons are the electrons in the outermost principal quantum level of an atom. Sometimes, the outermost energy level is called the valence shell. The outer energy level for this atom is n = 3, and it has two electrons in this energy level. Therefore, magnesium has two valence electrons.
Does B have 2 valence electrons?
Beryllium has two valence electrons. How many valence electrons does boron have? You must recognize that the second principal energy level consists of both the 2 s and the 2 p sublevels and so the answer is three.
How many valence electrons does a Mg2+ ion have?
if an atom of an element is electronically neutral then the number of protons (atomic number) will be equal to the number of electrons. Therefore Magnesium atom has 2 valence electrons, which explains why it likes to form Mg2+ cations by losing the latter 2 electrons.
Does magnesium have 8 valence electrons?
Now, magnesium, Mg , has 2 valence electrons. In order to complete its octet, it must get to 8 valence electrons. The only way for magnesium to accomplish this is to give up its two valence electrons and have one of its filled energy levels become the valence shell, i.e. the outermost energy level.
How many electron does magnesium have?
The most common and stable type of magnesium atom found in nature has 12 protons, 12 neutrons, and 12 electrons (which have a negative charge). Atoms of the same element with different neutron counts are known as isotopes.
What is the number of valence electrons in Na+?
In Na+ ion, there are only 10 electrons. The electronic configuration is 2 , 8. Therefore, the number of valence electrons in sodium ion is 8.
What is the valence of Na?
11 1
Valency of First 30 Elements
| Element | Atomic Number | Valency |
|---|---|---|
| Valency of Fluorine | 9 | 1 |
| Valency of Neon | 10 | 0 |
| Valency of Sodium (Na) | 11 | 1 |
| Valency of Magnesium (Mg) | 12 | 2 |
How many electrons does neon have?
2,8
Neon/Electrons per shell
What is the electron configuration for boron B?
[He] 2s2 2p1
Boron/Electron configuration
What is the number of electrons in Mg and Mg2+ ion?
Now, the atomic number is 12, so the number of electrons in a neutral magnesium atom is 12. Now, a magnesium atom has to lose 2 electrons in order to become Mg2+. So, it loses 2 electrons. Hence, the number of electrons in mg2+ ion will be 10.
What is the group number of magnesium?
2
Fact box
| Group | 2 | 650°C, 1202°F, 923 K |
|---|---|---|
| Atomic number | 12 | 24.305 |
| State at 20°C | Solid | 24Mg |
| Electron configuration | [Ne] 3s2 | 7439-95-4 |
| ChemSpider ID | 4575328 | ChemSpider is a free chemical structure database |