Is hydrogen bonding possible in ethanol?
Is hydrogen bonding possible in ethanol?
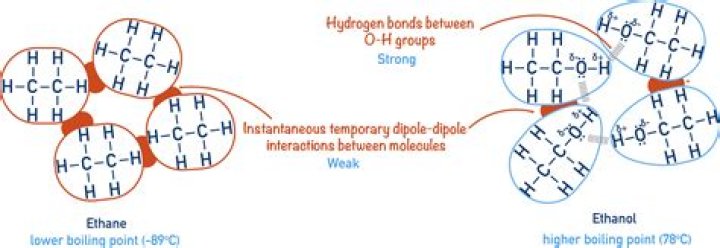
Hydrogen bonding can occur between ethanol molecules, although not as effectively as in water. The hydrogen bonding is limited by the fact that there is only one hydrogen in each ethanol molecule with sufficient δ+ charge.
Does ethanol ether have hydrogen bonding?
Ether molecules have no OH group and thus no intermolecular hydrogen bonding.
What type of bonding occurs in ethanol?
nonpolar covalent bonds
Ethanol, or C2H6O, has two different types of bonding between its constituent atoms. The bonds between the hydrogen and carbon atoms are nonpolar covalent bonds. The hydrogen-oxygen and carbon-oxygen bonds are polar covalent bonds.
Which type of hydrogen bonding is found in alcohol?
Answer: Methanol (CH3OH) forms intermolecular hydrogen bonds with other methanol molecules.
Why does the ability of ethanol to form hydrogen bonds results in ethanol have a higher boiling point than dimethyl ether?
Because hydrogen bonds are typically much stronger attractions than ordinary dipole moments, a group of ethanol molecules is much harder to separate from each other than a group of dimethyl ether molecules. The ethanol has a much higher boiling point.
What intermolecular forces exist between molecules of ethanol?
In ethanol, there is polar covalent bond between oxygen and hydrogen. Hence, there is dipole- dipole interaction in ethanol.
What is alcohol hydrogen bonding?
In the case of alcohols, hydrogen bonds occur between the partially-positive hydrogen atoms and lone pairs on oxygen atoms of other molecules. The hydrogen atoms are slightly positive because the bonding electrons are pulled toward the very electronegative oxygen atoms.
Is propanol a hydrogen bond?
The small alcohols ethanol, 1-propanol, and 2-propanol are miscible in water, form strong hydrogen bonds with water molecules, and are usually known as inhibitors for clathrate hydrate formation. However, in the presence of methane or other help gases, clathrate hydrates of these substances have been synthesized.
What is hydrogen bonding in alcohol?
Are alcohols hydrogen bond acceptors?
There are two H-bonding interactions for H-bond donors. Water and alcohols may serve as both donors and acceptors, whereas ethers, aldehydes, ketones and esters can function only as acceptors. Similarly, primary and secondary amines are both donors and acceptors, but tertiary amines function only as acceptors.
Why is the attraction between two molecules of ethanol stronger than the attraction between two molecules of dimethyl ether?
Because hydrogen bonds are typically much stronger attractions than ordinary dipole moments, a group of ethanol molecules is much harder to separate from each other than a group of dimethyl ether molecules.