Is tin a mixture
tin (Sn), a chemical element belonging to the carbon family, Group 14 (IVa) of the periodic table. It is a soft, silvery white metal with a bluish tinge, known to the ancients in bronze, an alloy with copper.
Is tin a mixture or compound?
Tin is a chemical element with the symbol Sn (from Latin: stannum) and atomic number 50.
How is tin classified?
Tin is the fourth element of the fourteenth column of the periodic table. It is classified as a post-transition metal. Tin atoms have 50 electrons and 50 protons with 4 valence electrons in the outer shell. Under standard conditions tin is a soft silvery-gray metal.
Is tin a compound?
Tin is a natural element in the earth’s crust. … Tin can combine with other chemicals to form compounds. Combinations with chemicals like chlorine, sulfur, or oxygen are called inorganic tin compounds (i.e., stannous chloride, stannous sulfide, stannic oxide).What is tin made of?
Tin is primarily obtained from the mineral cassiterite (SnO2) and is extracted by roasting cassiterite in a furnace with carbon. Tin makes up only about 0.001% of the earth’s crust and is chiefly mined in Malaysia. Two allotropes of tin occur near room temperature.
Is tin a semiconductor?
No, tin is not an elemental semiconductor. At room temperature, it is a conductor but below 55.8°F, it does not conduct electricity well.
Is a mixture of tin and copper?
Bronze is a mixture of copper and tin.
Is tin conductive?
Material IACS% ConductivityZinc27Brass28Iron17Tin15Is tin a pure substance?
Examples of pure substances include tin, sulfur, diamond, water, pure sugar (sucrose), table salt (sodium chloride) and baking soda (sodium bicarbonate). Crystals, in general, are pure substances. Tin, sulfur, and diamond are examples of pure substances that are chemical elements.
Is tin a metalloid?Other elements are occasionally classified as metalloids. These elements include hydrogen, beryllium, nitrogen, phosphorus, sulfur, zinc, gallium, tin, iodine, lead, bismuth, and radon.
Article first time published onHow is tin formed?
Fire refining is most commonly used and produces tin (up to 99.85 percent) suitable for general commercial use. Electrolytic refining is used on the products of complex ores and to produce a very high grade of tin (up to 99.999 percent). One fire-refining method is called boiling.
Is the element Sb a metal?
Antimony is a semi-metal. In its metallic form it is silvery, hard and brittle. Antimony is used in the electronics industry to make some semiconductor devices, such as infrared detectors and diodes.
Is tin radioactive element?
Tin (50Sn) is the element with the greatest number of stable isotopes (ten; three of them are potentially radioactive but have not been observed to decay), which is probably related to the fact that 50 is a “magic number” of protons. … The longest-lived radioisotope is 126Sn, with a half-life of 230,000 years.
How is tin refined?
Refining. There are two methods of refining impure tin. Fire refining is most commonly used and produces tin (up to 99.85 percent) suitable for general commercial use. Electrolytic refining is used on the products of complex ores and to produce a very high grade of tin (up to 99.999 percent).
How is tin formed naturally?
Sources of tin Tin is relatively rare, making up only about 2 parts per million of the Earth’s crust, according to the U.S. Geologic Survey. Tin is extracted from various ores, chiefly from Cassiterite (SnO2). The metal is produced from reducing the oxide ore with coal in a furnace.
What is ore of tin?
References. Cassiterite is a tin oxide mineral, SnO2. It is generally opaque, but it is translucent in thin crystals. Its luster and multiple crystal faces produce a desirable gem. Cassiterite was the chief tin ore throughout ancient history and remains the most important source of tin today.
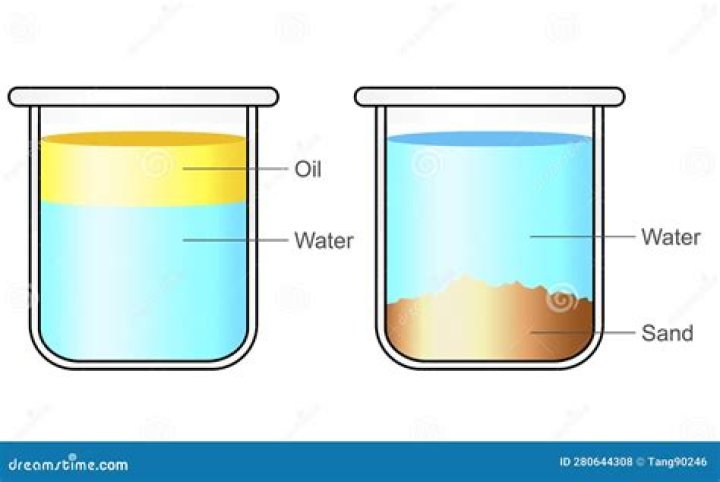
Is sand a mixture?
Sand is made up of particles of silicon dioxide as the main part (in different sizes). It is a heterogeneous mixture.
What is steel a mixture of?
Steel is an alloy of iron and carbon. Stainless steels are steels containing at least 10.5% chromium, less than 1.2% carbon and other alloying elements.
Is coal a mixture?
-Coal is made up of more than one compound so it is a mixture. … -Alcohol is made up of carbon, hydrogen, and oxygen so it is a compound. Note: Metals, non-metals, and metalloids are the classification of elements. Compounds are two types: organic compound and an inorganic compound.
Why tin is not a semiconductor?
Tin has a very large atomic radius hence the distance between valence electrons and nucleus is very large. Hence ionisation energy is very low such that the valence band and conduction band merge with each other.
Which element is a semiconductor?
Semiconductors are materials which have a conductivity between conductors (generally metals) and nonconductors or insulators (such as most ceramics). Semiconductors can be pure elements, such as silicon or germanium, or compounds such as gallium arsenide or cadmium selenide.
What is SB on the periodic table?
antimony (Sb), a metallic element belonging to the nitrogen group (Group 15 [Va] of the periodic table). … Antimony is a lustrous, silvery, bluish white solid that is very brittle and has a flaky texture.
Which of the following is a mixture?
Homogeneous mixtureHeterogeneous mixturecomposition and properties are uniform throughout the mixturecomposition and properties are non-uniform throughout the mixtureExample: Air, crude oil, ink, bloodExample: a mixture of sugar and sand, a mixture of wheat and rice
Is a mixture a pure substance?
a pure substance consists only of one element or one compound. a mixture consists of two or more different substances, not chemically joined together.
What are examples of pure substances and mixtures?
Pure SubstancesMixturesCan be categorised as elements and compounds.Can be categorized as only homogeneous and heterogeneous.Examples include Pure water, Hydrogen gas, Gold.Examples include a mixture of Sand and Sugar, Oil and Water etc.
Is a tin can a conductor or insulator?
Tin is a good conductor of electricity, insulators are non-conductive so it needs to be almost anything else besides a form of metal ferrous or non-ferrous.
Why is tin a good conductor?
It is these ‘free electrons’ that allow metals to conduct an electric current. Because valence electrons are free to move they can travel through the lattice that forms the physical structure of a metal. This physical structure is deferent with deferent metals and accordingly is electrical conductivity.
Is Tin an alloy?
tin (Sn), a chemical element belonging to the carbon family, Group 14 (IVa) of the periodic table. It is a soft, silvery white metal with a bluish tinge, known to the ancients in bronze, an alloy with copper.
Which is not metalloid?
Carbon is not a metalloid and is a non-metal. … The elements which are metalloids, in the periodic table are boron, silicon, arsenic, antimony, polonium, and tellurium. Metalloids are semiconductors.
Which element is a metalloid?
Survey of Elements Commonly Recognized as Metalloids The percentage appearance frequencies of the elements most frequently identified as metalloids are boron (86), silicon (95), germanium (96), arsenic (100), selenium (23), antimony (88), tellurium (98), polonium (49), and astatine (40).
What determines that an element is a metal?
A metal is generally lustrous, hard, malleable, ductile, and solid. It is a good conductor of heat and electricity. … Metals also form cations which are smaller than original atom. They mostly form ionic bonds although there are certain metals such as aluminium, iron, lithium and beryllium who form covalent bonds.