Is water a hydrogen or covalent bond
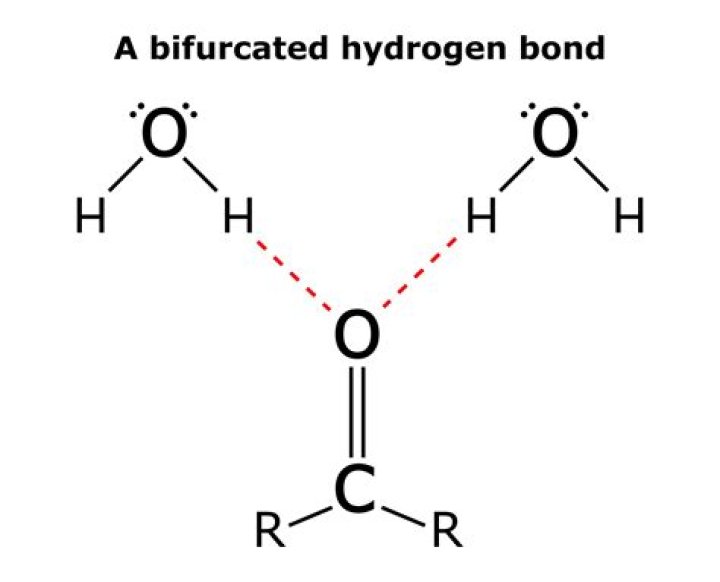
BIOdotEDU. A water molecule consists of two atoms of hydrogen linked by covalent bonds to the same atom of oxygen. Atoms of oxygen are electronegative and attract the shared electrons in their covalent bonds.
Is water a hydrogen bond?
Water. A ubiquitous example of a hydrogen bond is found between water molecules. In a discrete water molecule, there are two hydrogen atoms and one oxygen atom. The simplest case is a pair of water molecules with one hydrogen bond between them, which is called the water dimer and is often used as a model system.
Is hydrogen and water ionic or covalent?
PropertyIonicCovalentMelting temperatureHighLow
What type of bond is hydrogen and water?
In the case of water, hydrogen bonds form between neighboring hydrogen and oxygen atoms of adjacent water molecules. The attraction between individual water molecules creates a bond known as a hydrogen bond.Are hydrogen bonds covalent?
Hydrogen Bonding. Hydrogen bonding is a special type of dipole-dipole attraction between molecules, not a covalent bond to a hydrogen atom.
Why is H2O a covalent bond?
Water (H2O) is a covalent compound because the bond forms between two hydrogens and one oxygen are covalent in nature. The covalent bond is formed due to the sharing of electron occurs between hydrogen and oxygen atoms in order to complete their octet shell and hence, attains stability.
Why does hydrogen bonding occur in water?
In water molecules the oxygen atom attracts the negatively charged electrons more strongly than the hydrogen. This gives water an asymmetrical distribution of charge so that it is a polar molecule. … Because the water molecules are small, many of them can surround one molecule of the solute and form hydrogen bonds.
What type of molecule is water?
Water is a molecular compound consisting of polar molecules that have a bent shape. The oxygen atom acquires a partial negative charge, while the hydrogen atom acquires a partial positive charge.What are the bonds in a water molecule?
Strong linkages—called covalent bonds—hold together the hydrogen (white) and oxygen (red) atoms of individual H2O molecules. Covalent bonds occur when two atoms—in this case oxygen and hydrogen—share electrons with each other.
Is tap water molecular or ionic?Water is covalent compund,each of the 2 hydrogen atoms share 1 electron with oxygen and oxygen shares 2 electrons 1 with each hydrogen. But,now oxygen after sharing still have 2 lone pairs of electrons(electrons that have not participated in the formation of molecules). So,Water is polar covalent compound.
Article first time published onAre hydrophobic bonds covalent?
However the hydrophobic effect is not considered a non-covalent interaction as it is a function of entropy and not a specific interaction between two molecules, usually characterized by entropy.
What are the type of hydrogen bonding?
Hydrogen bondings are of two types, and it is classified as the following: The Intramolecular Hydrogen Bonding. The Intermolecular Hydrogen Bonding.
What is hydrogen bonding with example?
water (H2O): Water is an excellent example of hydrogen bonding. The bond is between the hydrogen of one water molecule and the oxygen atoms of another water molecule, not between the two hydrogen atoms (a common misconception). … ammonia (NH3): Hydrogen bonds form between hydrogen of one molecule and nitrogen of another.
Is H2O hydrogen bonding or dipole dipole?
The H2O water molecule is polar with intermolecular dipole-dipole hydrogen bonds. As the water molecules attract each other and form bonds, water displays properties such as high surface tension and a high heat of vaporization.
Is water is a covalent compound?
Water is a Polar Covalent Molecule The unequal sharing of electrons between the atoms and the unsymmetrical shape of the molecule means that a water molecule has two poles – a positive charge on the hydrogen pole (side) and a negative charge on the oxygen pole (side).
When a water molecule forms a hydrogen bond with another water molecule which of the following atoms are involved?
Two hydrogen atom from one molecule and one oxygen from the other molecule.
How many hydrogen bonds a water molecule can form with another water molecule?
Each water molecule can form two hydrogen bonds involving their hydrogen atoms plus two further hydrogen bonds utilizing the hydrogen atoms attached to neighboring water molecules. These four hydrogen bonds optimally arrange themselves tetrahedrally around each water molecule as found in ordinary ice (see right).
Why do hydrogen bonds form between water molecules quizlet?
Water molecules are made up of two hydrogen atoms and one oxygen atom. … Water molecules are polar which means that oxygen atoms are more electronegative than when compared to hydrogen atoms. Therefore, oxygen atoms attract to hydrogen atoms of other molecules, creating a weak hydrogen bond.
Is a water molecule water?
From a chemical perspective, water is a molecule in which a single oxygen atom is linked to two hydrogen atoms. It is less well known that water exists in two different forms (isomers) at the molecular level. The difference lies in the relative orientation of the nuclear spins of the two hydrogen atoms.
What is a hydrogen molecule?
A molecule of hydrogen is the simplest possible molecule. It consists of two protons and two electrons held together by electrostatic forces. Like atomic hydrogen, the assemblage can exist in a number of energy levels.
Is water an oxide of hydrogen?
Water is an oxide of hydrogen represented by the formula H2O.
Is a water an element?
water, a substance composed of the chemical elements hydrogen and oxygen and existing in gaseous, liquid, and solid states.
Are hydrogen bonds hydrophobic?
When a substance readily forms hydrogen bonds with water, it can dissolve in water and is referred to as hydrophilic (“water-loving”). Hydrogen bonds are not readily formed with nonpolar substances like oils and fats (Figure 1). These nonpolar compounds are hydrophobic (“water-fearing”) and will not dissolve in water.
Are hydrogen bonds hydrophobic or hydrophilic?
Nonpolar molecules that repel the water molecules are said to be hydrophobic; molecules forming ionic or a hydrogen bond with the water molecule are said to be hydrophilic.
Is water hydrophilic?
Water is a polar molecule that acts as a solvent, dissolving other polar and hydrophilic substances. In biology, many substances are hydrophilic, which allows them to be dispersed throughout a cell or organism. … This is caused by the attraction of water molecules to the hydrophilic molecules.
Can water form hydrogen bonds with phenyl groups?
Ben-Amotz and colleagues used Raman scattering to study benzene, phenol, and phenylalanine in water and found that liquid water does form hydrogen bonds with benzene and other aromatic rings, with the bonds forming with nearly every benzene molecule.
Are hydrogen bonds covalent or noncovalent?
Hydrogen bonds are another noncovalent bond. It is a type of attraction force between two atoms of two different molecules which is a weak attraction force.
Can water and carbon dioxide form a hydrogen bond?
CO2 can form hydrogen bonds with water, but its linear shape makes it a nonpolar molecule. This means that carbon dioxide is less soluble in water than polar molecules are. The solubility of carbon dioxide is increased when the water is cold, and decreased greatly when the water is warm.
Which of the following is not an example of hydrogen bond?
Liquid ammonia – contains Nitrogen, hence shows H-bonding. Water – contains Oxygen, hence shows H-bonding. Hydrochloric acid – does not contain Oxygen, Nitrogen or Fluorine, does not show hydrogen bonding. Therefore, the answer is – option (d) – Hydrochloric acid.
What is coordinate covalent bond with example?
An example of a dative covalent bond is provided by the interaction between a molecule of ammonia, a Lewis base with a lone pair of electrons on the nitrogen atom, and boron trifluoride, a Lewis acid by virtue of the boron atom having an incomplete octet of electrons.