Is water or dichloromethane more polar?
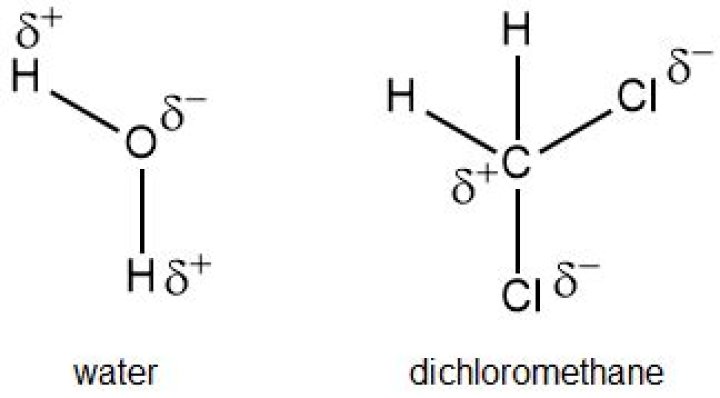
Is water or dichloromethane more polar?
Yeh. CH2Cl2 is barely polar actually since C-Cl has deltaEN=0.5 where C-H gives a 0.4. Much less polar than water for sure..
What is the polarity of dichloromethane?
| Solvent | Polarity Index | Dipole moment |
|---|---|---|
| Dichloromethane | 3.1 | 1.14 |
| 1,2-Dichloroethane | 3.5 | 1.83 |
| Isopropyl Alcohol | 3.9 | 1.66 |
| n-Butyl Acetate | 4.0 | 1.84 |
Which of the following solvents is most polar?
In the give molecules water is the most polar molecule due to its higher ionic character and large difference in electronegativity. Hexane is an organic molecule with no heteroatoms; hence it will be the least polar solvent.
Which of the following solvents is the least polar?
hexane
Note that hexane, which is 100% hydrocarbon, is the least polar solvent in the table.
Is water soluble in dichloromethane?
Dichloromethane (DCM or methylene chloride) is an organochloride compound with the formula CH2Cl2. This colorless, volatile liquid with a chloroform-like, sweet odour is widely used as a solvent….Dichloromethane.
| Names | |
|---|---|
| Solubility in water | 25.6 g/L (15 °C) 17.5 g/L (25 °C) 15.8 g/L (30 °C) 5.2 g/L (60 °C) |
Why is dichloromethane polar and nonpolar?
Dichloromethane is polar because it has different polarity bonds and its shape cannot arrange those bond dipoles to cancel out. Note that polarity can be considered for the whole molecule, functional group or particular bonds. E.g. CO2 has zero permanent dipole moment, as bond dipoles cancel each other.
Is dichloromethane soluble in water?
It is moderately soluble in water (2 g/100 ml at 20 °C) and soluble in most organic solvents such as ethanol, ether, phenols, aldehydes and ketones. Its evaporation rate is 27.5 (reference liquid is butyl acetate = 1). DCM vapours are heavier than air.
Why is dichloromethane not soluble in water?
Organic solvents like dichloromethane are immiscible in water because water is, by contrast, a very polar solvent. However, DCM is actually denser than water, and leaves an organic layer below the aqueous layer rather than above like other solvents.
Is water polar or nonpolar?
Water is a Polar Covalent Molecule The unequal sharing of electrons between the atoms and the unsymmetrical shape of the molecule means that a water molecule has two poles – a positive charge on the hydrogen pole (side) and a negative charge on the oxygen pole (side).
Is DCM polar or nonpolar?
Dichloromethane (DCM or methylene chloride) is an organochloride compound with the formula CH2Cl2. This colorless, volatile liquid with a chloroform-like, sweet odour is widely used as a solvent. Although it is not miscible with water, it is polar, and miscible with many organic solvents.
Why is dichloromethane insoluble in water?
Organic solvents like dichloromethane are immiscible in water because water is, by contrast, a very polar solvent. When non-polar substances enter an aqueous solution, they exhibit hydrophobic behaviour by aggregating together and excluding the water molecules.