What are 3 major types of bonds?
What are 3 major types of bonds?
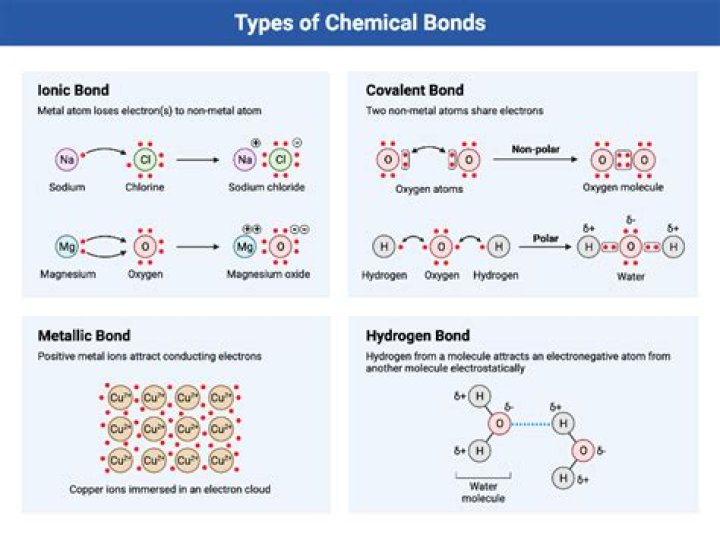
There are many types of chemical bonds that can form, however the 3 main types are: ionic, covalent, and metallic bonds.
How do you identify bond types?
Identifying Types of Bonds
- Look at the chemical formula.
- Identify the elements in the compound.
- Determine if the elements are metals or nonmetals (using a periodic table)
- Metal – Metal = Metallic.
- Metal – Nonmetal = Ionic.
- Nonmetal — Nonmetal = Covalent.
What are the different types of bonds explain?
Ionic bonds form when a nonmetal and a metal exchange electrons, while covalent bonds form when electrons are shared between two nonmetals. An ionic bond is a type of chemical bond formed through an electrostatic attraction between two oppositely charged ions.
How do the three types of bonds differ?
The three different types of bonding are covalent, ionic and metallic bonding. Ionic bonding occurs between metal and non-metal atoms. It involves the transfer of electrons from the metal to the non-metal producing a positively charged metal ion and negatively charged non-metal ion.
What are the types of bonding?
There are three primary types of bonding: ionic, covalent, and metallic.
- Ionic bonding.
- Covalent bonding.
- Metallic bonding.
What is bond and its types?
There are three main types of bonds: Corporate bonds are debt securities issued by private and public corporations. Investment-grade. These bonds have a higher credit rating, implying less credit risk, than high-yield corporate bonds. High-yield.
What are 2 types of bonds?
Ionic bonds form when a nonmetal and a metal exchange electrons, while covalent bonds form when electrons are shared between two nonmetals.
What kind of bond is N and O?
covalent bonds
The bonds between nitrogen and oxygen are covalent bonds made from sharing electron pairs.