What are biological oxidation reactions?
What are biological oxidation reactions?
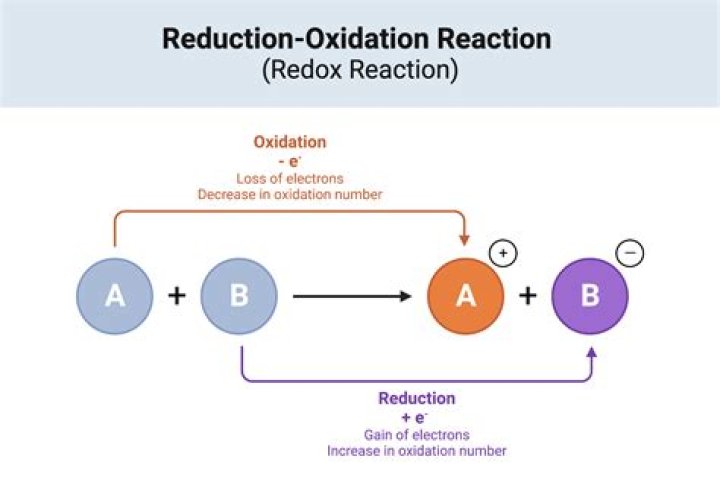
Biological oxidation is the combination of oxidation-reduction transformations of substances in living organisms. Oxidation-reduction reactions are those which take place with a change in the oxidation state of atoms through the redistribution of electrons between them.
What is oxidation reaction in organic compounds?
In reference to organic molecules, oxidation is a process by which a carbon atom gains bonds to more electronegative elements, most commonly oxygen. Reduction is a process by which a carbon atom gains bonds to less electronegative elements, most commonly hydrogen.
What is an example of biological oxidation?
As the magnesium is oxidized there is a loss of 2 electrons while simultaneously, oxygen gains those two electrons. Another example of a redox reaction is with the two gasses CO2 and H2.
What is biological oxidation-reduction reaction?
An oxidation is defined as loss of electrons in the course of a chemical reaction. If a species gains electrons, it is undergoing a reduction. Since electrons are “conserved” in a chemical reaction (they are not created or destroyed),one chemical species’ loss is another’s gain.
Where is biological oxidation occur?
The second, more complex method of transforming nutrients into energy is anaerobic biological oxidation, or tissue respiration. This reaction takes place in all aerobic organisms which use oxygen in the respiration process.
What enzymes are involved in biological oxidation?
Enzymes involved in oxidation and reduction are called oxidoreductases and are classified into four groups: oxidases, dehydrogenases, hydroperoxidases, and oxygenases. Oxidases use oxygen as a hydrogen acceptor.
Why does the oxidation of organic compounds?
Why does the oxidation of organic compounds by molecular oxygen to produce CO2 and water release free energy? The oxidation of organic compounds releases less energy than the energy stored in the covalent bonds of CO2 and water. The covalent bond in O2 is unstable and easily broken by electrons from organic molecules.
What organic compounds can be oxidized?
Alcohols may be oxidized to give ketones, aldehydes, and carboxylic acids. These functional groups are useful for further reactions; for example, ketones and aldehydes can be used in subsequent Grignard reactions, and carboxylic acids can be used for esterification. Oxidation of organic compounds generally…
Where does biological oxidation occur?
What are the enzymes involved in biological oxidation?
What is oxidation reaction with example?
The term oxidation was originally used to describe reactions in which an element combines with oxygen. Example: The reaction between magnesium metal and oxygen to form magnesium oxide involves the oxidation of magnesium.
What is an example of an oxidation-reduction reaction?
The tarnishing of silver is just one example of a broad class of oxidation-reduction reactions that fall under the general heading of corrosion. Another example is the series of reactions that occur when iron or steel rusts. When heated, iron reacts with oxygen to form a mixture of iron(II) and iron(III) oxides.