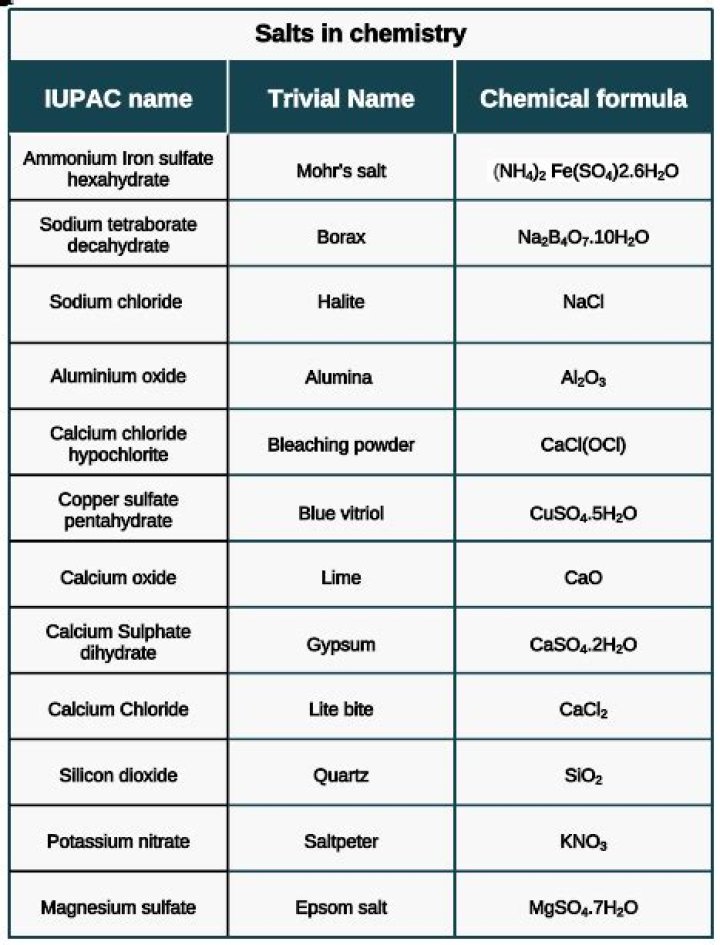
What are salts in chemistry
salt, in chemistry, substance produced by the reaction of an acid with a base. A salt consists of the positive ion (cation) of a base and the negative ion (anion) of an acid. … The term salt is also used to refer specifically to common table salt, or sodium chloride.
What are examples of salts in chemistry?
- Sodium Chloride. ••• Sodium chloride (NaCl) is the most common type of salt in our lives. …
- Potassium Dichromate. ••• Potassium dichromate (K2Cr2O7) is an orange-colored salt composed of potassium, chromium and oxygen. …
- Calcium Chloride. ••• …
- Sodium Bisulfate. ••• …
- Copper Sulfate. •••
Which are salts?
Salts are ionic compounds which, when dissolved in water, break up completely into ions. They arise by the reaction of acids with bases, and they always contain either a metal cation or a cation derived from ammonium (NH4+). Examples of salts include NaCl, NH4F, MgCO3, and Fe2(HPO4)3.
How do you identify a salt in chemistry?
- Look at the Appearance of the Compound. Typically the compound appears in a solid form. …
- Check the Effect of Heating. …
- Conduct a Flame Test. …
- Test Its Reaction with Hydrochloric Acid. …
- Note Its Solubility in Water. …
- Sieve analysis. …
- Moisture analysis.
What are the five types of salt in chemistry?
- Normal salt : A salt that does not contain any replaceable hydrogen atoms or hydroxyl groups is called normal salt. …
- Thus, an acid salt may be defined as follows.
What are the seven salts?
- Table salt. …
- Sea salt. …
- Black sea salt (Hawaiian sea salt) …
- Pink Himalayan salt. …
- Persian Blue Salt. …
- Sel gris and fleur de sel. …
- Smoked salts.
Are all salts salty?
Not all salts are safe to eat, and not all of them taste salty. The cation determines if a salt has a salty flavour, and the anion determines the intensity of that flavour. To interact with our taste receptors, salts first have to split back – or dissociate – into their ions.
What is salt analysis?
Salt analysis is the quantitative and qualitative analysis of cations and anions present in a given inorganic salt. The salt analysis involves a series of steps that help in identifying a salt and its constituent ions. … Cations can be classified into six different groups according to the properties that they possess.What are salts in chemistry class 10?
Salts: Salts are the ionic compounds which are produced after the neutralization reaction between acid and base. Salts are electrically neutral. There are number of salts but sodium chloride is the most common among them. Sodium chloride is also known as table salt or common salt.
What is the PH of salt?Here NaCl is a Neutral base,and hence the PH is equal to 7.
Article first time published onWhat is salt Short answer?
In chemistry, a salt is any neutral chemical compound made of cations (positive ions) attached to anions (negative ions). The main kind of salt is sodium chloride with the chemical formula NaCl. It is formed when hydrochloric acid is added in sodium hydroxide.
What is a salt chemistry GCSE?
A salt is a compound formed when some or all the hydrogen ions in an acid are replaced by metal ions or ammonium ions. For example, zinc sulfate is the salt formed when zinc ions replace the hydrogen ions of sulfuric acid.
What are the 10 examples of salts?
- Sodium Chloride or Common Salt (NaCl)
- Sodium Hydroxide (NaOH)
- Sodium Carbonate or Washing Soda (Na2CO3.10H2O)
- Baking Soda or Sodium Bi-carbonate (NaHCO3)
- Bleaching Powder or Calcium Hypochlorite.
- Plaster of Paris or Hemihydrate Calcium sulphate, CaSO4 1/2 H2O.
What are the 4 types of salt?
- Kosher salt. Kosher salt is a coarse-grained flaky salt. …
- Table salt. This is the most common type of salt, and the one most used in home kitchens. …
- Sea salt. Sea salt refers to unrefined salt that is sourced from — appropriately — the sea. …
- Coarse salt.
What are the basic salts?
Alkali salts or basic salts are salts that are the product of incomplete neutralization of a strong base and a weak acid. … Another definition of a basic salt would be a salt that contains amounts of both hydroxide and other anions. White lead is an example. It is basic lead carbonate, or lead carbonate hydroxide.
How are salts made?
salt, in chemistry, substance produced by the reaction of an acid with a base. A salt consists of the positive ion (cation) of a base and the negative ion (anion) of an acid. The reaction between an acid and a base is called a neutralization reaction.
Is there tasteless salt?
We have emphasized that in its natural form, without additives, salt doesn’t lose its saltiness or flavor. Consumable salt is a mineral compound consisting of sodium and chloride (NaCI). It is extremely stable and therefore cannot lose its savor or degrade over time, unlike spices.
What is the taste of salt called?
The simplest receptor found in the mouth is the sodium chloride (salt) receptor. Saltiness is a taste produced primarily by the presence of sodium ions. Other ions of the alkali metals group also taste salty, but the further from sodium, the less salty the sensation is.
Are ions salt?
Salt is an ionic compound, consisting of a crystal, lattice structure of the two ions Na+ and Cl-.
Is HCl a salt?
Hydrochloric acid is the salt of the protonated water and chloride. Its ions are often written as H3O+ Cl−, although the cation is in fact often bonded to other water molecules.
Are all salts bases?
Keep in mind that a salt will only be basic if it contains the conjugate base of a weak acid. Sodium chloride, for instance, contains chloride (Cl–), which is the conjugate base of HCl. But because HCl is a strong acid, the Cl– ion is not basic in solution, and it isn’t capable of deprotonating water.
What is the rarest salt?
Salt. Amabito No Moshio (Seaweed Salt, also referred to as Ancient Sea salt) is perhaps the world’s rarest and most expensive sea salt and produced only in Japan.
How are salts formed Class 7?
A salt is formed when a metal displaces hydrogen from an acid.
What are neutral salts Class 10?
Neutral Salt: Salts produced because of reaction between strong acid and strong base are neutral in nature. The pH value of such salts is equal to 7, i.e. neutral. Example; Sodium chloride, sodium sulphate, potassium chloride, etc.
What is acid bases and salt?
An acid is defined as a substance whose water solution tastes sour, turns blue litmus red and neutralizes bases. A substance is called base if its aqueous solution tastes bitter, turns red litmus blue or neutralizes acids. Salt is a neutral substance whose aqueous solution does not affect litmus.
What are three types of salts?
Three types of salts are neutral, acidic and basic salts. The salts that give a neutral solution on mixing and dissolving them with water are known as neutral salts. The reaction of strong acid and strong base gives neutral salt. For example sodium chloride, sodium sulphate.
What is cation and anion?
Cations are positively-charged ions (atoms or groups of atoms that have more protons than electrons due to having lost one or more electrons). Anions are negatively-charged ions (meaning they have more electrons than protons due to having gained one or more electrons).
Why are salts classified into groups?
The different cations are classified under six groups depending upon the difference in the solubilities of their chlorides, sulphide hydroxides and carbonates at different pH values.
What is the pH of blood?
The acidity and alkalinity of your blood are measured using the pH scale. The pH scale ranges from 0 (very acidic) to 14 (very alkaline). Blood is usually between 7.35 to 7.45.
What is pH full form?
The letters pH stand for potential of hydrogen, since pH is effectively a measure of the concentration of hydrogen ions (that is, protons) in a substance. The pH scale was devised in 1923 by Danish biochemist Søren Peter Lauritz Sørensen (1868-1969).
Is salt acid or alkaline?
In addition to water, a new compound is formed from the rest of the acid and alkali molecule. This is known as a salt. In chemistry, a salt is a compound formed by an acid and an alkali. In everyday English though, the word refers to only one particular type of salt: Sodium chloride or table salt.