What are the 2 hydrogen isotopes?
What are the 2 hydrogen isotopes?
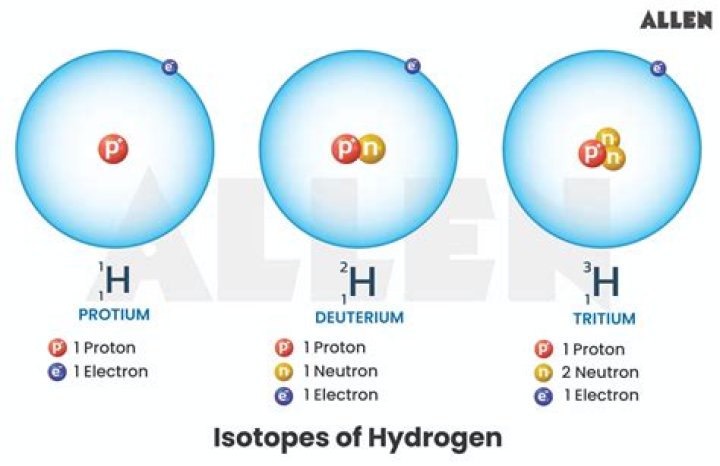
Hydrogen is the only element whose isotopes have different names that remain in common use today: the 2H (or hydrogen-2) isotope is deuterium and the 3H (or hydrogen-3) isotope is tritium. The symbols D and T are sometimes used for deuterium and tritium.
Which is the most common isotope of hydrogen?
Protium
Protium, 1H, has no neutrons in its nucleus and is the most common form of hydrogen, with an atomic mass of ~1.0078 Da (dalton) and an isotopic abundance of ~99.972% of all hydrogen on Earth.
What is hydrogen 2 used for?
The Hydrogen isotope H-2, also known as deuterium, is used in a variety of applications. Deuterium is used extensively in organic chemistry in order to study chemical reactions. It is also used in vitamin research.
How do the isotopes hydrogen 1 and hydrogen-2 differ?
Hydrogen-2 has one neutron; hydrogen-1 has none. Hydrogen-2 has two protons; hydrogen-1 has one.
What are isotopes write the isotopes of hydrogen?
How many isotopes are in hydrogen? The hydrogen element has three isotopes: hydrogen, deuterium, and tritium. We each have a single proton (Z = 1), but the number of their neutrons is different. There is no neutron in hydrogen, one in deuterium, and two neutrons in tritium.
What are the three types of hydrogen isotopes?
There are three isotopes of the element hydrogen: hydrogen, deuterium, and tritium. How do we distinguish between them? They each have one single proton (Z = 1), but differ in the number of their neutrons. Hydrogen has no neutron, deuterium has one, and tritium has two neutrons.
What is the importance of heavier isotopes of hydrogen?
an atom of tritium has one proton and two neutrons is its nucleus. tritium atoms are unstable and are radioactive whereas the former two are non-radioactive. It is found in heavy water. heavy water is used to control the nuclear reaction in nuclear reactor and helps to slow down the speed of neutrons.
What is the H isotope with 1p and 2n?
The nucleus of deuterium, called a deuteron, contains one proton and one neutron (mass number = 2), whereas the far more common hydrogen isotope, protium, has no neutrons in the nucleus.
What does the number 84 in the name krypton 84 represent?
In the case of Krypton-84, this means that you have 84 nucleons, where 36 of these are protons, and the remaining 48 are neutrons. This becomes relevant in nuclear chemistry, where sometimes, you have to balance nuclear equations with respect to the mass numbers (nucleon numbers).