What are the difference between these isotopes of hydrogen?
What are the difference between these isotopes of hydrogen?
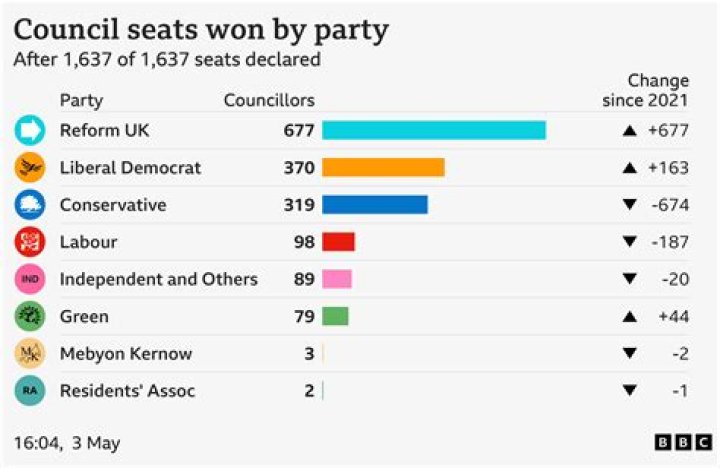
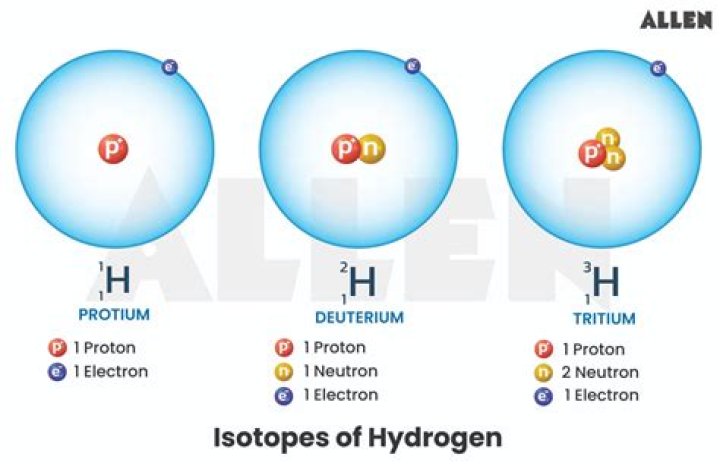
Each isotope of hydrogen is made up of only one proton, but these isotopes differ in the number of neutrons the atoms contain. The extra neutrons make the isotopes heavier: deuterium is twice as heavy as ordinary hydrogen (sometimes called protium), while tritium is three times as heavy.
What is the difference between hydrogen 1 and hydrogen-2?
Protium (hydrogen-1) has an atomic mass of 1.00782504, and is a stable isotope. It has one proton and no neutrons. Deuterium (hydrogen-2) is the second most abundant isotope of hydrogen and it makes up 0.0026 to 0.0184% of the hydrogen that is naturally found on the Earth.
What are the 3 stable isotopes of hydrogen?
- The three most stable isotopes of hydrogen: protium (A = 1), deuterium (A = 2), and tritium (A = 3).
- Protium, the most common isotope of hydrogen, consists of one proton and one electron.
- A deuterium atom contains one proton, one neutron, and one electron.
What is the difference between hydrogen deuterium and tritium?
Deuterium and tritium are isotopes of hydrogen, the most abundant element in the universe. Whereas all isotopes of hydrogen have one proton, deuterium also has one neutron and tritium has two neutrons, so their ion masses are heavier than protium, the isotope of hydrogen with no neutrons.
How do the isotopes of hydrogen-2 and hydrogen 3 differ?
The image shows the three isotopes of the element hydrogen. All three forms have one proton (pink) and one electron (dark green) but differ in the number of neutrons (gray) in the nucleus. Deuterium, or hydrogen-2 (bottom left) has one neutron. Tritium, or hydrogen-3 (bottom right) has two neutrons.
What is the difference between hydrogen 3 and Helium 3?
Helium-3 contains two protons and one neutron, while tritium contains one proton and two neutrons.
How do the isotopes of hydrogen-2 and hydrogen-3 differ?
What is the difference between hydrogen-3 and Helium 3?
How do the isotopes hydrogen-2 and hydrogen 3 differ?
All three forms have one proton (pink) and one electron (dark green) but differ in the number of neutrons (gray) in the nucleus. Protium, or ordinary hydrogen (top), has no neutrons. Deuterium, or hydrogen-2 (bottom left) has one neutron. Tritium, or hydrogen-3 (bottom right) has two neutrons.
What are the three isotopes of hydrogen and how do they differ quizlet?
From left to right, the isotopes are protium (1H) with zero neutrons, deuterium (2H) with one neutron, and tritium (3H) with two neutrons. Isotopes are variants of a particular chemical element which differ in neutron number, although all isotopes of a given element have the same number of protons in each atom.
Where do isotopes differ?
Isotopes are different forms of the same element that are different from each other according to the number of neutrons they have in their nuclei.
What is the difference between he 3 and he-4?
The quantum mechanical effects on helium-3 and helium-4 are significantly different because with two protons, two neutrons, and two electrons, helium-4 has an overall spin of zero, making it a boson, but with one fewer neutron, helium-3 has an overall spin of one half, making it a fermion.