What are the different types of chemical bonding with example?
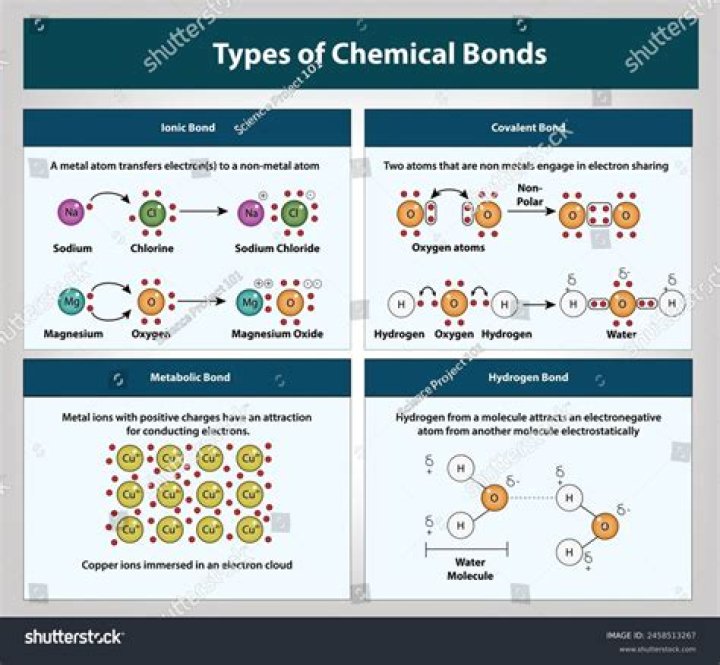
What are the different types of chemical bonding with example?
Different Types of Chemical Bonds with Examples
- Ionic Bond. As the name suggests, ionic bonds are a result of the attraction between ions.
- Covalent Bond. In the case of a covalent bond, an atom shares one or more pairs of electrons with another atom and forms a bond.
- Hydrogen Bond.
- Metallic Bonds.
What are the main types of chemical bonds How are they different from one another?
The two main types of bonds formed between atoms are ionic bonds and covalent bonds. An ionic bond is formed when one atom accepts or donates one or more of its valence electrons to another atom. A covalent bond is formed when atoms share valence electrons.
What is covalent bond and examples?
Examples of compounds that contain only covalent bonds are methane (CH4), carbon monoxide (CO), and iodine monobromide (IBr). Covalent bonding between hydrogen atoms: Since each hydrogen atom has one electron, they are able to fill their outermost shells by sharing a pair of electrons through a covalent bond.
How many types of chemical bonds are there in chemistry?
There are four types of bonds or interactions: ionic, covalent, hydrogen bonds, and van der Waals interactions. Ionic and covalent bonds are strong interactions that require a larger energy input to break apart.
What is chemical Bonding Class 10?
A chemical bond is a formation of bond due to the electrostatic forces of attraction due to sharing of electrons or donating electrons. The attractive force which holds up the constituent particles (atoms, ions or molecules) together in a chemical species is known as chemical bond.
What are 2 types of chemical bonds?
Covalent and ionic bonds are both typically considered strong bonds. However, other kinds of more temporary bonds can also form between atoms or molecules.