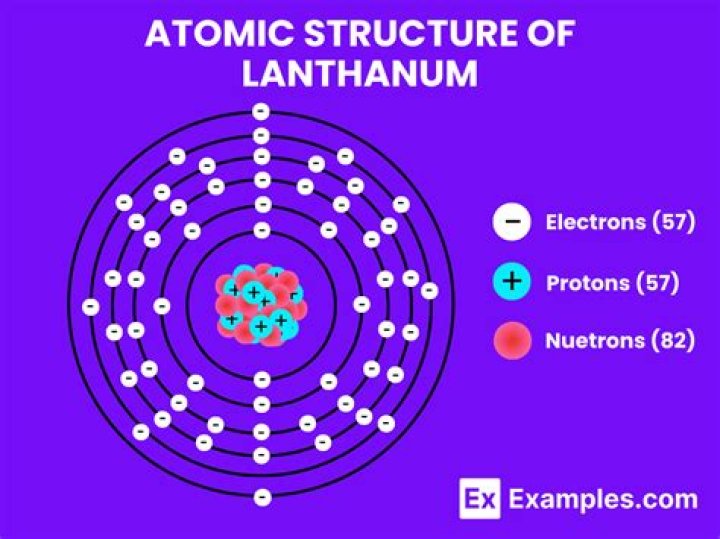
What are the electrons in the outermost energy orbitals?
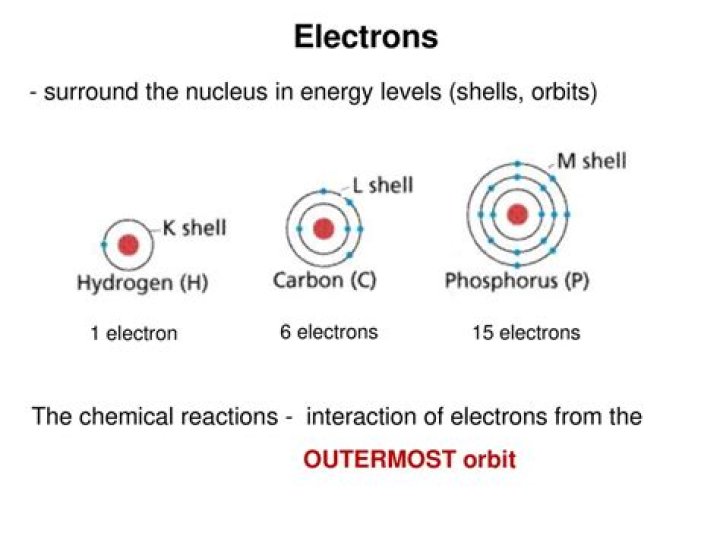
What are the electrons in the outermost energy orbitals?
Electrons in the outermost energy level of an atom are called valence electrons.
How is an atom’s outer electrons energy levels filled?
Thus, the electron shells of an atom are populated from the inside out, with electrons filling up the low-energy shells closer to the nucleus before they move into the higher-energy shells further out.
Is there a full outer electron level?
Group 18 elements (helium, neon, and argon are shown) have a full outer, or valence, shell. A full valence shell is the most stable electron configuration. Elements in other groups have partially-filled valence shells and gain or lose electrons to achieve a stable electron configuration.
In what order do electrons fill energy levels?
The order of increasing energy of orbitals as shown below, is backed by experimental data. The arrangement of electrons within an atom is called the electronic configuration and the electrons are filled up according to the energy of the levels as: 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, 6s, 4f, 5d, 6p, 7s, 5f.
What is the outermost energy level?
valence shell
The outermost energy level that (in the ground state of the atom) contains electrons is called the “valence shell” of the atom. These valence electrons are responsible for all chemical bonds between atoms. For silicon, the valence shell corresponds to a principle quantum number n of 3.
Does the third energy level hold 8 or 18 electrons?
The third shell in its lowest state has room for 8 electrons but including the higher energy 3d electrons it has a capacity of 18 electrons.
What is Group 18 on the periodic table called?
noble gas, any of the seven chemical elements that make up Group 18 (VIIIa) of the periodic table. The elements are helium (He), neon (Ne), argon (Ar), krypton (Kr), xenon (Xe), radon (Rn), and oganesson (Og).
Why is 3rd shell 8 or 18?
The third shell of an atom has 18 electrons only not 8 electrons. You might be confused because first the electrons of 4s are filled and then the 10 electrons of 3d shells are filled. They are filled because of the n-l rule.
Which 20 elements have full outer shells of electrons?
The group (family) with elements cotaining full outer shells are the right-most group on the table: the noble gases: helium, neon, argon, krypton, xenon, and radon (element 118, oganesson, also belongs in this group, but most all of its chemical and physical properties are as of yet unknown.
How do electrons fill in orbitals?
According to the principle, electrons fill orbitals starting at the lowest available energy states before filling higher states (e.g., 1s before 2s). The Madelung energy ordering rule: Order in which orbitals are arranged by increasing energy according to the Madelung Rule.
In which of the following orbitals electron will be filled first?
4s orbitals
We say that the 4s orbitals have a lower energy than the 3d, and so the 4s orbitals are filled first. We know that the 4s electrons are lost first during ionization. The electrons lost first will come from the highest energy level, furthest from the influence of the nucleus.