What are the main reactions shown by carbonyl compounds?
What are the main reactions shown by carbonyl compounds?
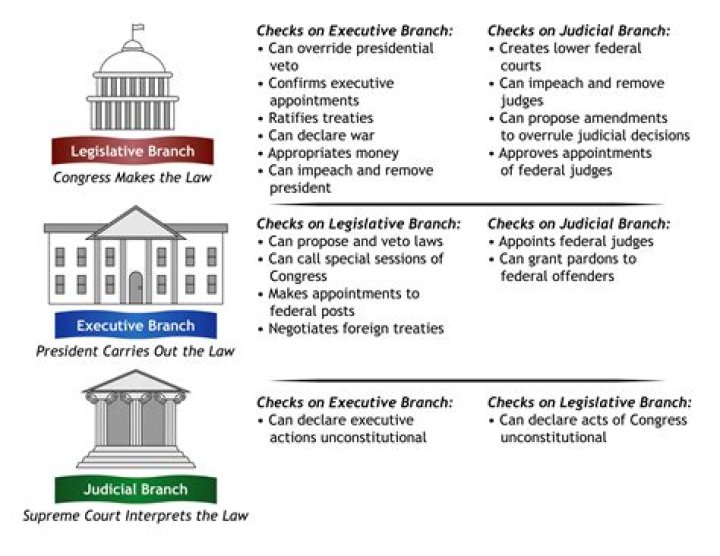
Carbonyl compounds show the following types of chemical reactions :
- Nucleophilic addition reactions.
- Nucleophilic addition-elimination reactions.
- Oxidation, reduction, and halogenation.
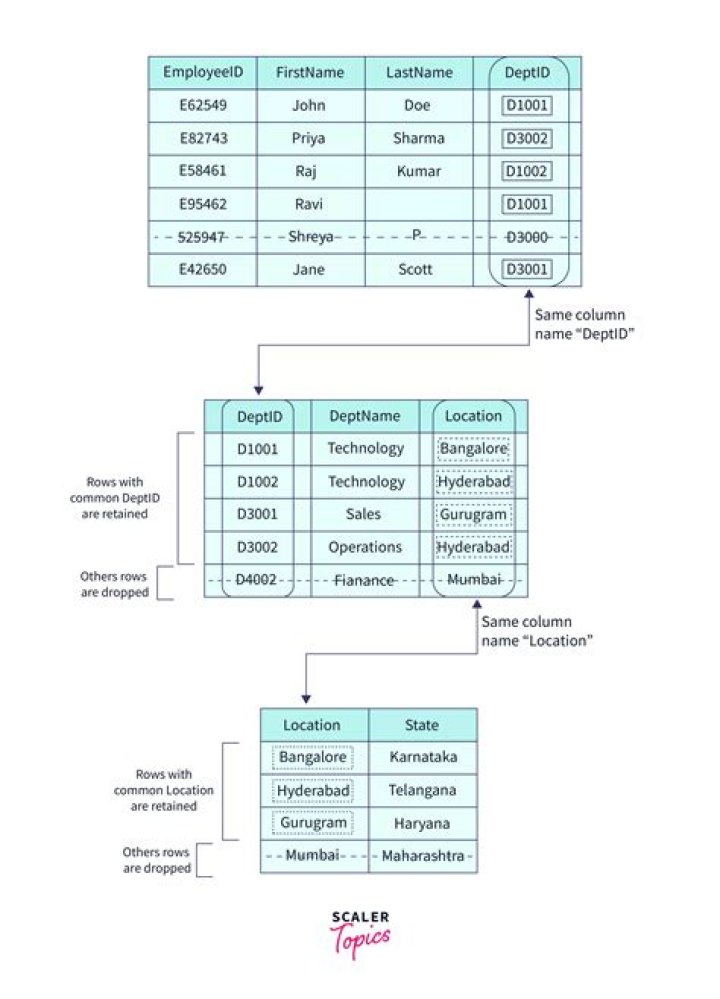
Which reactions can be used to prepare carbonyl compounds?
Following are some important methods of preparation of aldehydes.
- By Oxidation of Alcohol. After the oxidation of primary and secondary alcohols we can get both aldehydes and ketones.
- Dehydrogenation of Alcohols. This is widely used in industries.
- From Hydrocarbons.
- By Oxidation of Methylbenzene.
What are the two common reagents used in the reduction of carbonyl containing compounds?
Of the two most common reducing reagents, LiAlH4 is the more powerful, reducing most carbonyl containing compounds including aldehydes, ketones, esters, and amides. A more selective reducing agent is NaBH4, which only reacts with aldehydes and ketones due to its milder nature.
What happens when hydrazine reacts with carbonyl compounds?
Reaction of carbonyl with hydrazine gives a hydrazone. Hydrazine is more nucleophilic than a regular amine due to the presence of the adjacent nitrogen. Note the similarity to the formation of an oxime reaction. Hydrazones generally form a mixture of geometric isomers.
What is carbonyl reaction?
The reactions of carbonyls can be broadly classified as the direct nucleophilic addition reactions wherein a nucleophile adds to the carbonyl carbon atom. The other equally important and versatile family of reactions of carbonyl arise due to the acidity of the alpha-C-H groups.
What are carbonyl compounds give two examples?
Examples of inorganic carbonyl compounds are carbon dioxide and carbonyl sulfide. A special group of carbonyl compounds are 1,3-dicarbonyl compounds that have acidic protons in the central methylene unit. Examples are Meldrum’s acid, diethyl malonate and acetylacetone.
What are carbonyl compounds give method of preparation?
1. Aldehydes and ketones can be prepared by oxidation of primary and secondary alcohols. 2. Oxidation of alcohols involves the formation of a Carbon – Oxygen double bond with cleavage of an O-H and C-H bonds.
Which class of compound is an example of a carbonyl compound?
Examples of class 1 carbonyl compounds are amides, esters, carboxyllic acids and anhydrides. Class 2 compounds have carbonyl compounds that do not leave the molecule when reacting, examples of this type of carbonyl compound are ketones and aldehydes.
What reagent is used for reduction of alcohol to carbonyl compound?
Reduction of Aldehydes and Ketones The most common sources of the hydride Nucleophile are lithium aluminum hydride (LiAlH4) and sodium borohydride (NaBH4). Note! The hydride anion is not present during this reaction; rather, these reagents serve as a source of hydride due to the presence of a polar metal-hydrogen bond.
What happens when carbonyl compounds are treated with hydrazine write the equation?
Answer: The reduction of aldehydes and ketones to alkanes. Condensation of the carbonyl compound with hydrazine forms the hydrazone, and treatment with base induces the reduction of the carbon coupled with oxidation of the hydrazine to gaseous nitrogen, to yield the corresponding alkane.
How does benzaldehyde react with phenylhydrazine?
Benzaldehyde reacts with phenylhydrazine to give a hydrazone, demonstrating a chemical reaction in which C=O. bond breaking is involved. Strong coupling is at the heart of optomechanics where it enables coherent quantum state transfer between light and micromechanical oscillators.
What are the carbonyl compounds?
In organic chemistry, a carbonyl group is a functional group composed of a carbon atom double-bonded to an oxygen atom: C=O. It is common to several classes of organic compounds, as part of many larger functional groups. A compound containing a carbonyl group is often referred to as a carbonyl compound.