What does aspartic acid do in catalytic triad?
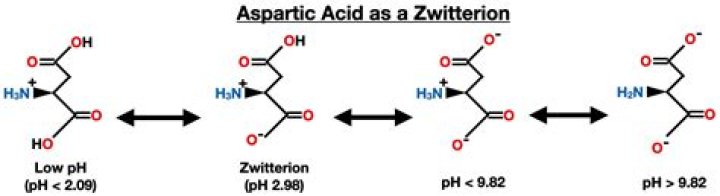
What does aspartic acid do in catalytic triad?
A catalytic triad charge-relay system as commonly found in proteases. The acid residue (commonly glutamate or aspartate) aligns and polarises the base (usually histidine) which activates the nucleophile (often serine or cysteine, occasionally threonine).
What is aspartic protease?
Aspartic proteases are one of the four classes of proteolytic enzymes, which cut other proteins into smaller pieces. Proteolytic enzymes are also known as peptidases, because they cleave peptide bonds, and as proteinases, because they cleave proteins.
What forms the catalytic triad in the serine protease?
9 Catalytic Triad. The serine esterases have a catalytic triad: serine, glutamic or aspartic acid, and histidine. These catalytic residues are responsible for the nucleophilic attack of the active site serine on the carbonyl carbon atom of the ester.
What are the types of proteases?
Based on the mechanism of catalysis, proteases are classified into six distinct classes, aspartic, glutamic, and metalloproteases, cysteine, serine, and threonine proteases, although glutamic proteases have not been found in mammals so far.
What is the pH of protease?
The proteases with pH optima in the range of 2.0–5.0 are called acid proteases, proteases having pH optima around 7.0 are neutral proteases, and alkaline proteases have pH optima in the range of 8.0–11.0.
Where do aspartic proteases cleave?
Catalytic mechanism Aspartyl proteases are a highly specific family of proteases – they tend to cleave dipeptide bonds that have hydrophobic residues as well as a beta-methylene group.
What is the role of aspartate in serine proteases?
Abstract. The role of the aspartic acid residue in the serine protease catalytic triad Asp, His, and Ser has been tested by replacing Asp102 of trypsin with Asn by site-directed mutagenesis. The catalytic activity of the mutant enzyme increases with increasing pH so that at pH 10.2 the kcat is 6 percent that of trypsin …
What is the difference between a serine protease and an aspartate protease?
Aspartic proteases are a group of protease enzymes that use two highly conserved aspartic acid residues in the active site for catalytic cleavage of their peptide substrates. Unlike serine or cysteine proteases these proteases do not form a covalent intermediate during cleavage.
Is serine protease a hydrolase?
Serine hydrolases are one of the largest known enzyme classes comprising approximately ~200 enzymes or 1% of the genes in the human proteome. Superfamilies of serine hydrolases includes: Serine proteases, including trypsin, chymotrypsin, and subtilisin.
Is protease a proteolytic enzyme?
proteolytic enzyme, also called protease, proteinase, or peptidase, any of a group of enzymes that break the long chainlike molecules of proteins into shorter fragments (peptides) and eventually into their components, amino acids.