What does p-nitrophenol do?
What does p-nitrophenol do?
p-Nitrophenyl Phosphate (PNPP) is a non-proteinaceous, non-specific substrate used to assay protein, alkaline and acid phosphatases. p-Nitrophenyl Phosphate (PNPP) is a non-proteinaceous, non-specific substrate used to assay protein, alkaline and acid phosphatases.
Why does p-nitrophenol turn yellow?
pH indicator The yellow color of the 4-nitrophenolate form (or 4-nitrophenoxide) is due to a maximum of absorbance at 405 nm (ε = 18.3 to 18.4 mM−1 cm−1 in strong alkali).
What is p-nitrophenol PNP?
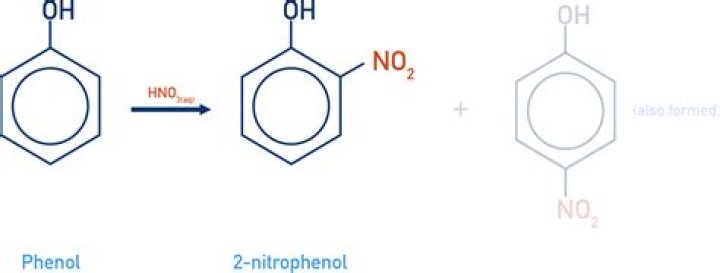
para-Nitrophenol (PNP) is a common environmental pollutant owing to its wide application in pharmaceuticals, explosives, dyes and agrochemicals. PNP also accumulates in the soil due to the hydrolysis of organophosphorus insecticides such as parathion or methyl parathion (MP) [1].
What is the application of p-nitrophenol in spectrophotometry?
The acyl esters of p-nitrophenol are the most commonly used model substrates to study the esterolytic activity of hydrolases. A quantitative detection of the released p-nitrophenolate can be easily carried out at 405–410 nm by spectrophotometry (Schmidt & Bornscheuer, 2005).
What color is p-nitrophenol?
Colorless Yellow
More videos on YouTube
| Indicator | Color at pH incidated | |
|---|---|---|
| Below 6 | 6-9 | |
| p-nitrophenol | Colorless | Yellow |
| Phenolphthalein | Colorless | Colorless |
| (mixture of phenolphthalein and p-nitrophenol) | Colorless | Yellow |
Is 4-nitrophenol soluble in water?
4-Nitrophenol is a colorless to light yellow solid with very little odor. 2-Nitrophenol is slightly soluble in cold water, but 4-nitrophenol is moderately soluble in cold water.
Is Pnpp a good substrate?
Thermo Scientific Pierce PNPP (p-nitrophenyl phosphate) is a colorimetric, soluble substrate of alkaline phosphatase for use in ELISA applications. PNPP (p-Nitrophenyl Phosphate, Disodium Salt) is a widely used substrate for detecting alkaline phosphatase in ELISA applications.
Is 4-nitrophenol toxic?
Poisonous by ingestion and moderately toxic by skin contact. Acute (short-term) inhalation or ingestion of 4-nitrophenol in humans causes headaches, drowsiness, nausea, and cyanosis (blue color in lips, ears, and fingernails).
What do spectrophotometer readings mean?
Spectrophotometry is a measurement of how much a chemical substance absorbs or transmits. With the spectrophotometer, the amount of a known chemical substance (concentrations) can also be determined by measuring the intensity of light detected.
What Colour is pNPP?
pNPP (p-nitrophenylphosphate) is a chromogenic substrate for Alkaline Phosphatase (AP). In the presence of AP, pNPP is hydrolyzed rapidly to p-nitrophenol and inorganic phosphate. Liberated p-nitrophenol is seen as a clear yellow color that is measured at 405/620 nm.