What does the Suzuki reaction do?
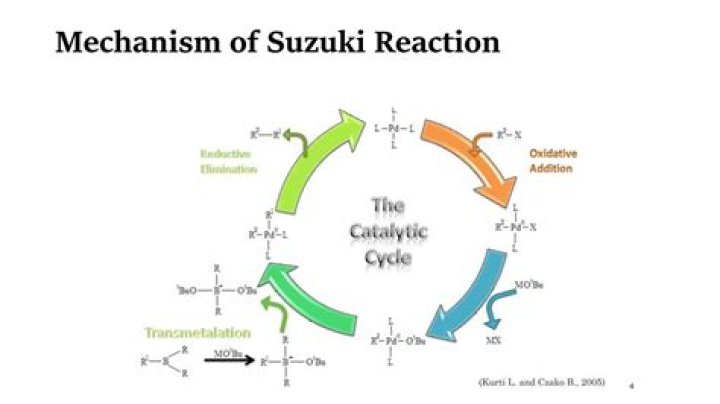
What does the Suzuki reaction do?
Suzuki reaction can conjoin a variety of aryl halides and alkenyl halides with alkenylboranes and arylboronic acids and is thus an essential method of synthesizing many styrenes, alkenes, and biphenyls. It is very cost-effective for use in the synthesis of intermediates for pharmaceuticals or fine chemicals.
What is a boronic ester?
Boronic esters (RB(OR)2), also referred to as boronate esters, are formed between a boronic acid and an alcohol. With their excellent ability to construct C-C bonds via metal-catalyzed cross coupling, boronic esters have received much attention for use in organic synthesis.
What is the role of water in Suzuki reaction?
These water molecules form as a by-product of a side reaction, the trimerization of phenylboronic acid. This small amount of water can play a key role in the desired transformation, making it a pseudo-solid-state reaction. The team reacted solid aryl halides with phenylboronic acid in the absence of any liquids.
What is a Boronate?
Noun. boronate (plural boronates) (organic chemistry) Any salt or ester of a boronic acid.
Is Suzuki reaction air sensitive?
Reaction Conditions: Milder and Greener Organoboranes are nontoxic and stable to extreme heating and exposure to oxygen or water. Consequently, these reagents can be easily used at benchtop, and do not require special equipment or techniques, such as gloveboxes or air-sensitive and dry technique.
Why palladium is used in Suzuki reaction?
It also allows this reaction to proceed at lower temperatures (20 °C from the usual 50 to 80 °C). Unfortunately, these bases are air and light sensitive, but are still widely used. Palladium catalysts are the most widely used for Suzuki coupling and perform best with electron-donating (usually phosphine) ligands.
What is the role of base in Suzuki coupling?
Table 3. Effect of the catalyst and the base on the selectivity in the Suzuki reaction between two boronic acids. [equiv.] [a] All reactions were performed in H2O/DMF (1:1) with 1 equiv.
How do you make an Eoronic Ester?
Boronic esters (also named boronate esters) Boronic esters are esters formed between a boronic acid and an alcohol. The compounds can be obtained from borate esters by condensation with alcohols and diols.