What element is Xe 6s2 5d1?
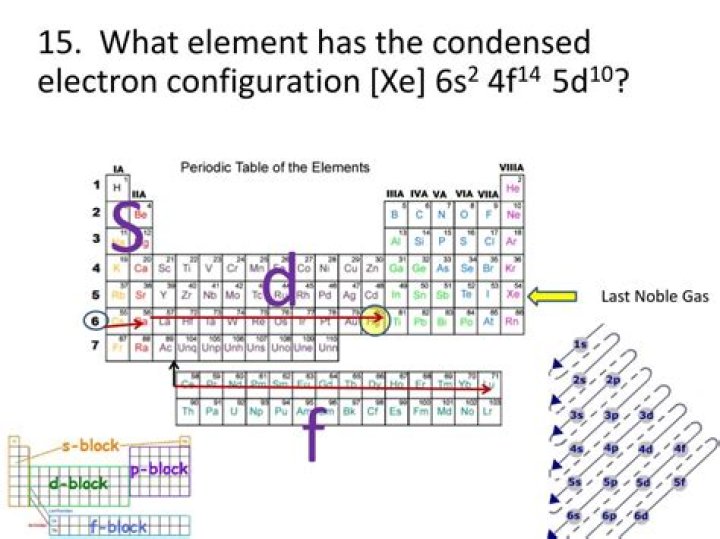
What element is Xe 6s2 5d1?
Electron Configuration Chart – Electron Configuration of all the elements in table chart
| Element Atomic Number | Element Symbol | Element Electron Configuration |
|---|---|---|
| 55 | Cs | [Xe] 6s1 |
| 56 | Ba | [Xe] 6s2 |
| 57 | La | [Xe] 5d1 6s2 |
| 58 | Ce | [Xe] 4f1 5d1 6s2 |
What is XE 4f1 5d1 6s2?
The ground state electronic configuration of Neutral Cerium atom is [Xe] 4f1 5d1 6s2. The portion of Cerium configuration that is equivalent to the noble gas of the preceding period, is abbreviated as [Xe]. For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.
Why is lanthanum 5d1 6s2?
As the number of energy levels and sublevels increases it becomes complex enough so that the energy levels overlap. By the addition of one electron to the 5d raises the energy of the level so that the next electrons go to 4f. Hence Lanthanum [Xe] 5d1 6s2 instead of [Xe] 6s2 4f1.
In what group of the periodic table will the element found having electronic configuration Xe 4f 14 5d4 6s2?
Explanation: 4f14 5d0 6fs2 belongs to f–block, III B group.
What is XE 6s2?
Barium has the noble gas configuration [Xe]6s².
What does Xe mean in electron configuration?
| Xenon | |
|---|---|
| Group | group 18 (noble gases) |
| Period | period 5 |
| Block | p-block |
| Electron configuration | [Kr] 4d10 5s2 5p6 |
What element ends with 4f1?
Cerium would end in 4f1, Pr in 4f2. These rare earth metals are 2 periods behind because the f electrons are even higher in energy than the d electrons.
What group does cerium belong to?
Lanthanides
Fact box
| Group | Lanthanides | Melting point |
|---|---|---|
| Period | 6 | Boiling point |
| Block | f | Density (g cm−3) |
| Atomic number | 58 | Relative atomic mass |
| State at 20°C | Solid | Key isotopes |
Why is 5d1 filled before 4f1?
The 4f sub shell and 5d sub shell has almost same energy level so it is tought to tell the diference. This Lanthanide contraction explains why 5s and 5p orbitals Penetrate the 4f sub-shell and filling of electron takes place in 5d sub shells instead of in 4f subshell.
Is lanthanum a 5d1?
The electron configuration of lanthanum is 6s2 5d1 but the electron configuration of cerium is 6s2 4f2. So from lanthanum to cerium you take one electron out of the d-orbital and put it in the second f-orbital.
Where is the element with the electronic configuration Xe 5d1 located in the periodic table?
It belongs to group 3 of the periodic table since all f-block elements belong to group 3. Its electronic configuration is [Xe] 4f7 5d1 6s2. Thus, its atomic number is 54 + 7 + 2 + 1 = 64. Hence, the element is Gadolinium.
What is the electronic configuration of Xe?
[Kr] 4d¹⁰ 5s² 5p⁶
Xenon/Electron configuration