What happen when lead nitrate is heated?
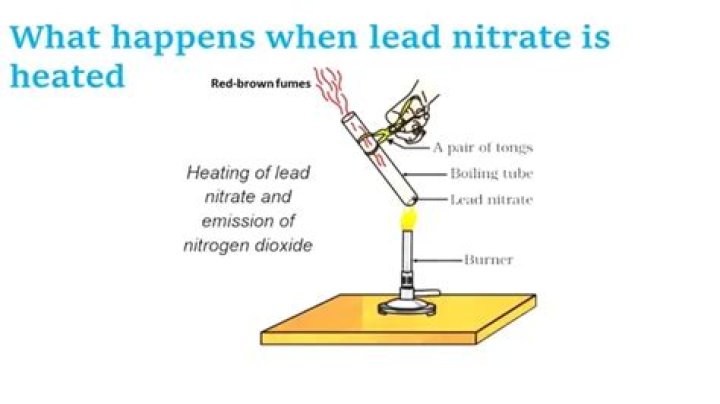
What happen when lead nitrate is heated?
When lead nitrate is heated it decomposes to form lead oxide, nitrogen dioxide and oxygen.
What is the Colour of lead nitrate when heated?
When lead nitrate is heated in air, it decomposes to give lead oxide, a reddish brown coloured nitrogen dioxide gas and oxygen.
What happens when lead nitrate heat and write the reaction?
On heating lead nitrate decomposition reaction takes place and lead nitrate get splits into lead oxide,nitrogen dioxide and oxygen gas. When lead(II) nitrate is heated, it decomposes into lead (II) oxide, nitrogen dioxide and oxygen.
What happens when lead nitrate is heated in a test tube?
Lead nitrate, on heating strongly in a dry test tube, produces Lead oxide(PbO), Nitrogen dioxide( NO2) and oxygen(O2). The lead nitrate appears to turn yellow due to the resultant product- lead oxide(PbO) and the evolution of a reddish-brown gas which is nitrogen dioxide.
When lead nitrate is heated a yellow solid residue of?
lead oxide
Explanation: When solid lead nitrate is heated, a yellow residue (lead oxide) is obtained, along with brown colored gas (nitrogen dioxide), and oxygen is liberated.
What color does lead oxidize?
When we apply heat over the lead oxide it changes its colour to yellow. This newly generated yellow colored oxide of lead is naturally unstable and as the temperature falls down it again changes into its stable form which is red in colour. Yellow colored oxide of lead is also known as meta-stable oxide.
When lead nitrate is heated which gas is evolved?
Answer:When lead nitrate is heated strongly it breaks down to form simpler substances like lead monoxide,nitrogen dioxide and oxygen.In this decomposition reaction the colourless compound lead nitrate forms a yellow compound lead monoxide and brown fumes of nitrogen dioxide gas are evolved.
What do you observe when a boiling test tube containing lead nitrate powder is heated it over a flame?
Upon heating lead nitrate strongly, it decomposes with a crackling sound and a precipitate of lead oxide is formed along with the release of oxygen and nitrogen dioxide gas.
Which products will be formed when lead nitrate is heated?
Answer: When lead nitrate is heated it decomposes to form a lead oxide, nitrogen dioxide and oxygen. Thus, lead nitrate on decomposition gives lead oxide, nitrogen dioxide and oxygen.
What gas is evolved when lead nitrate is strongly heated?
Nitrogen dioxide
Detailed Solution. Nitrogen dioxide is the reddish brown gas evolved when Lead Nitrate is heated strongly. Lead nitrate on heating decomposes to lead monoxide, nitrogen dioxide and oxygen.
What is the colour of lead residue?
Lead carbonate is a white coloured powder when heated it forms a red coloured residue. This residue is called litharge or lead monoxide which is lead (II)oxide.
Is lead nitrate colorless?
Lead(II) nitrate is an inorganic compound with the chemical formula Pb(NO3)2. It commonly occurs as a colourless crystal or white powder and, unlike most other lead(II) salts, is soluble in water.



