What happens below critical micelle concentration?
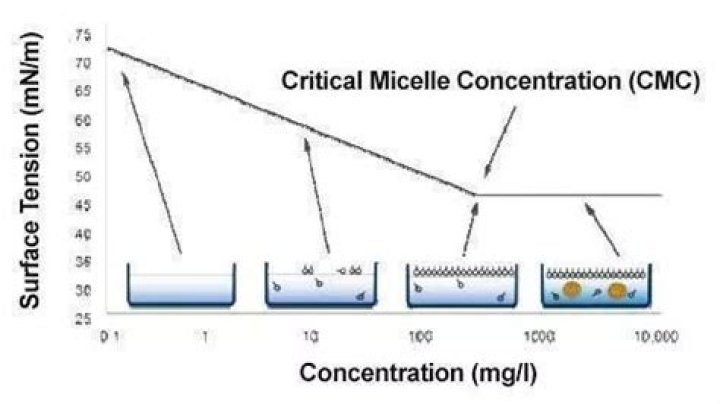
What happens below critical micelle concentration?
Below the CMC point, interfacial tension between oil and water phase is no longer effectively reduced. If the concentration of the surfactant is kept a little above the CMC, the additional amount covers the dissolution with existing brine in the reservoir.
What is the critical micelle concentration of CTAB?
0.0009 mol/L
The CMC of CTAB is 0.0009 mol/L in water, while it is 0.24 mol/L in ethanol. Furthermore, the dissipative particle dynamics (DPD) was adopted to simulate the aggregation of CTAB in water and ethanol/water mixtures, and the energy difference was calculated for the surfactant tail groups after mixing with the solvent.
What is micelle theory?
Definition of micellar theory : a theory in cytology: protoplasm and some of its products (as the plant cell wall) exist primarily as or are largely made up of micelles.
What are the effects of CMC?
There are several factors affecting the CMC point of a surfactant. These include the amphiphile chain length, dissolved salts, the structure of the head group, temperature, the structure of the alkyl chain and polar additives.
What is CMC explaining CMC factors?
Size and structure of hydrophobic group: An increase in the hydro-carbon chain length causes a logarithmic decrease in the CMC. Nature of hydrophilic group: An increase in hydrophilicity increases the CMC due to increased surfactant solubility in the aqueous medium and reduced partitioning into the interface.
How do you find the critical micelle concentration on a graph?
The Critical Micelle Concentration (CMC) can be measured using the intrinsic viscosity. This concentration is determined by ploting the specific viscosity as a function of the overlap parameter; the CMC corresponds to the chages of the slop in this graph.
What is the role of Krafft temperature in micelle formation?
Krafft temperature is defined as the minimum temperature from which the micelle formation takes place. Below the Krafft temperature, the maximum solubility of the surfactant will be lower than the critical micelle concentration, meaning micelles will not form.
What are the two conditions required for the formation of micelles?
Micelles form only when the concentration of surfactant is greater than the critical micelle concentration (CMC), and the temperature of the system is greater than the critical micelle temperature, or Krafft temperature.
What are types of micelles?
Micelles are usually spherical ranging in size from 2 to 20 nm depending on composition. The most common methods for micelle preparation include oil in water emulsion [98,99], solvent evaporation [100,101], solid dispersion, and dialysis methods [97,102].
What CMC means?
Computer-mediated communication
Computer-mediated communication (CMC) is defined as any human communication that occurs through the use of two or more electronic devices.
What is CMC in food?
Carboxymethyl cellulose (CMC) is a sodium salt derivative of cellulose. CMC finds use in gluten-free baking by providing dough with viscosity and bread with volume much like gluten proteins do. It also functions well in fillings as a thickener and in glazes as an agent to slow down sugar crystallization.
What is the critical micelle concentration CMC and why is it important to know?
The CMC (critical micelle concentration) is the concentration of a surfactant in a bulk phase, above which aggregates of surfactant molecules, so-called micelles, start to form. The CMC is an important characteristic for surfactants.