What happens when aldehyde reacts with semicarbazide?
What happens when aldehyde reacts with semicarbazide?
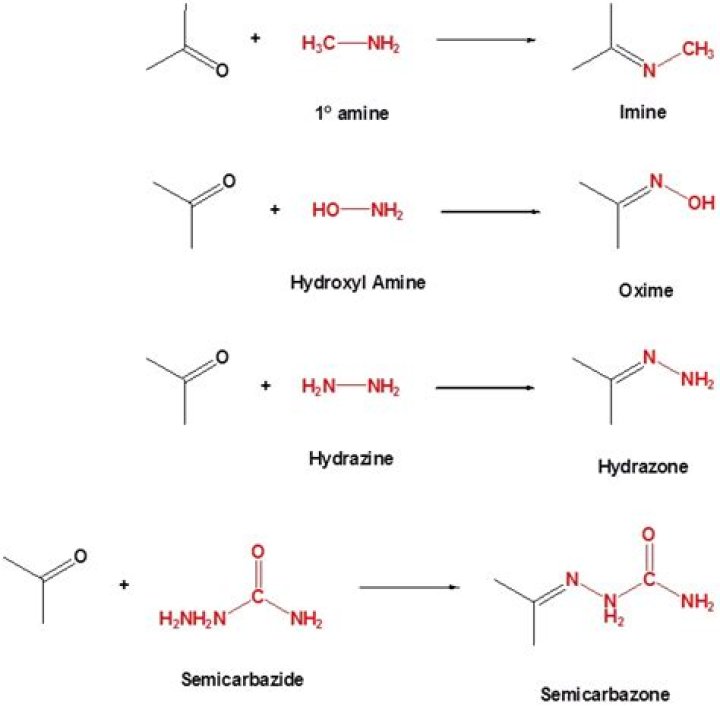
Derivatives. Semicarbazide is frequently reacted with aldehydes and ketones to produce semicarbazones via a condensation reaction. The reaction is useful because semicarbazones, like oximes and 2,4-DNPs, typically have high melting points and crystallize, facilitating purification or identification of reaction products …
How do aldehydes react with tollens reagent?
Tollens’ reagent oxidizes an aldehyde into the corresponding carboxylic acid. Ketones are not oxidized by Tollens’ reagent, so the treatment of a ketone with Tollens’ reagent in a glass test tube does not result in a silver mirror (Figure 1; right).
Does phenyl aldehyde give tollens test?
Yes,The hydroxyl amines on warning with ammoniacal silver nitrate solution (Tollen’s reagent) get oxidised to nitroso compound and reduce the Tollens reagent to metallic silver. This reaction is used as a test for nitro compounds and is called Baker – Mullikens’ test.
What happens when an aldehyde is treated with Tollen’s reagent and with Fehling’s solution?
The reaction requires heating of aldehyde with Fehling’s Reagent which will result in the formation of a reddish-brown colour precipitate. Hence, the reaction results in the formation of carboxylate anion. However, aromatic aldehydes do not react to Fehling’s Test. Moreover, ketones do not undergo this reaction.
What is the formula of semicarbazide?
CH5N3O
Semicarbazide/Formula
Which of the following is a GHS hazard code for semicarbazide hydrochloride?
CAS 563-41-7, EC Number 209-247-0, chemical formula CH₅N₃O * HCl. H301: Toxic if swallowed….Pricing & Availability.
| Product Information | |
|---|---|
| Molar Mass | 111.53 g/mol |
| HS Code | 2928 00 90 |
| Quality Level | MQ200 |
What is Tollen’s reagent formula?
1 . The Chemical formula for Tollen’s reagent is [ Agno3 + Nh4 oh ] . It is Same as [Ag (Nh3)2] OH .
When aldehyde oxidizes with Tollen’s reagent what is precipitate out in reaction?
Aldehyde gives a grey black precipitate or a silver mirror when freshly prepared Tollens’ reagent is added to the solution. The Basic Procedure Followed as: The small amount (50mg) of the given compound is dissolved in an aldehyde free alcohol (2ml-acting as a neutral solvent).
Does formaldehyde give Tollens test?
Formaldehyde and acetaldehyde both have alpha hydrogen. The compound with alpha hydrogen will show a positive Tollens test. Thus, both the given aldehydes will show a positive Tollens test.
What is Tollen’s reagent and Fehling’s solution?
Tollen’s test: this test is used to differentiate between a ketone and an aldehyde. In this test aldehyde is oxidised where as the ketone will not undergo oxidation. Fehling’s test: This test is used to detection of reducing sugar. It contain two solution Fehling’s solution A and Fehling’s solution B.
Which pair can be distinguished by using Tollen’s reagent?
Tollen’s test is used to distinguish between propionaldehyde and acetone because Tollen’s reagent is a weak oxidising agent and results in the formation of silver mirror with aldehydes.
What is semicarbazide derivative?
In organic chemistry, a semicarbazone is a derivative of imines formed by a condensation reaction between a ketone or aldehyde and semicarbazide.