What happens when you add HCl to an alkene?
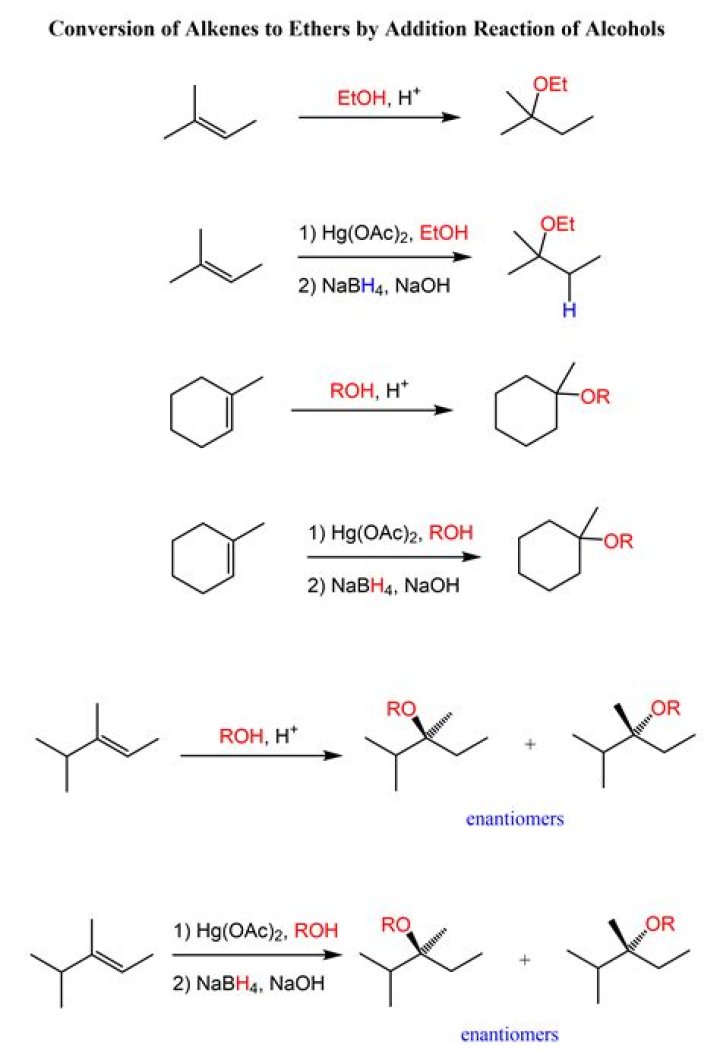
What happens when you add HCl to an alkene?
In a reaction with a polar molecule such as hydrogen chloride (HCl), for example, the π bond of an alkene reacts as a nucleophile. Thus, the partially positively charged hydrogen atom of HCl acts as an electrophile protonating the double bond of the alkene. The reaction type is A + B → C, an addition reaction.
What isomer will be the major product of the addition of HCl to 1 butene?
Addition of HCl to 3-methyl-1-butene gives 2-chloro-2-methyl butane as the major product.
How is ethanol produced from an alkene?
Manufacturing alcohols from alkenes. Ethanol is manufactured by reacting ethene with steam. The catalyst used is solid silicon dioxide coated with phosphoric(V) acid. The reaction is reversible.
What happens when alkene reacts with alcohol?
Since the dehydration reaction of alcohol has a carbocation intermediate, hydride or alkyl shifts can occur which relocates the carbocation to a more stable position. The dehydrated products therefore are a mixture of alkenes, with and without carbocation rearrangement.
What is the product when 1 butene reacts with HCl?
The addition of HCl to 3,3-dimethyl-1-butene, for example, leads to an unexpected product, 2-chloro-2,3-dimethylbutane, in somewhat greater yield than 3-chloro-2,2-dimethylbutane, the expected Markovnikov product.
What does HCl do in a reaction?
HCl + CH3OH → CH3O+H2 + Cl. Hydrogen chloride can protonate molecules or ions and can also serve as an acid-catalyst for chemical reactions where anhydrous (water-free) conditions are desired. Because of its acidic nature, hydrogen chloride is a corrosive substance, particularly in the presence of moisture.
How is ethanol manufactured?
The steps in the ethanol production process include milling the corn to meal, liquefying the meal by adding water and cooking, breaking down starch into sugar, using yeast to ferment the sugar to ethanol, distilling the ethanol by boiling off and condensing it by removing residual water and finally denaturing so that …
How is ethanol industrially produced?
Ethanol is manufactured by reacting ethene with steam. The catalyst used is solid silicon dioxide coated with phosphoric(V) acid. By removing the ethanol from the equilibrium mixture and recycling the ethene, it is possible to achieve an overall 95% conversion.
How is ethanol produced by fermentation and what’s the by product of this reaction?
When yeast is added it feeds on the sugar in the absence of oxygen to form wine (a solution of ethanol) and carbon dioxide. A chemical reaction called fermentation takes place in which the glucose is broken down to ethanol by the action of enzymes in the yeast.
How many products are possible when 2 butene reacts with HCl?
In the reaction between 2-pentene and HCl, two products will be form and names of the products will be 2-chloropentane and 3-chloropentane.
Is the reaction of 1 ethyl 2 Methylcyclohexene with HCl a regioselective reaction explain?
Question: Is the reaction of 1-ethyl-2-methylcyclohexene with HCl a regioselective reaction? Yes, as the sp carbon bonded to the ethyl group is more stable since there are more atoms in an.