What is a polytropic process in thermodynamics?
What is a polytropic process in thermodynamics?
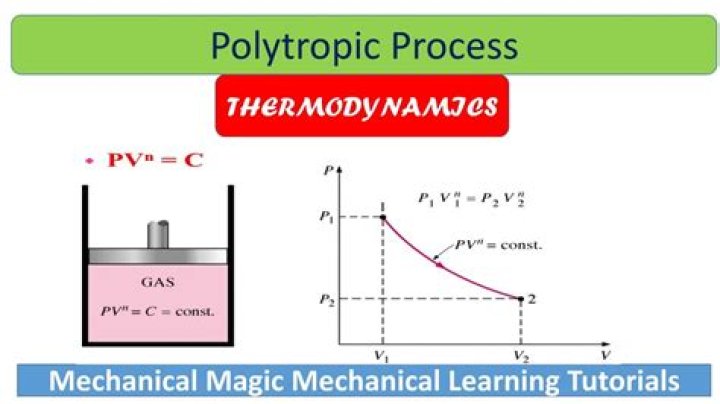
A polytropic process is a thermodynamic process that obeys the relation: where p is the pressure, V is volume, n is the polytropic index, and C is a constant. The polytropic process equation can describe multiple expansion and compression processes which include heat transfer.
What are the thermodynamic processes for the Otto cycle *?
The four-stroke Otto cycle is made up of the following four internally reversible processes: 1–2, isentropic compression; 2–3, constant-volume heat addition; 3–4, isentropic expansion; and 4–1, constant-volume heat rejection.
What happen during Polytropic process?
The term “polytropic” was originally coined to describe any reversible process on any open or closed system of gas or vapor which involves both heat and work transfer, such that a specified combination of properties were maintained constant throughout the process.
Is a polytropic process isothermal?
Isothermal process are a subset of the polytropic. Polytropic encompass all other common processes and more. standard notation implying C is a constant, P the pressure and V the volume.
Is Polytropic process adiabatic?
PVn = constant Where P is the pressure, V is the volume and n is a constant. Hence, to hold PV constant in the polytropic gas expansion/compression process, both heat and work interchange takes place between the system and surrounding. Therefore, polytropic is a non-adiabatic process.
What thermodynamic cycle do cars use?
the Otto Cycle
Specifically, the vast majority of automobiles seen on the road today use the Otto Cycle to convert gasoline into motion. Any machine (the list would go on and on) that uses gasoline will be divided into two categories of engines as seen below.
What is Brayton cycle in thermodynamics?
The Brayton cycle is a thermodynamic cycle that describes the operation of certain heat engines that have air or some other gas as their working fluid. It is also sometimes known as the Joule cycle. The reversed Joule cycle uses an external heat source and incorporates the use of a regenerator.
Which of the following is true for a polytropic process?
7. Which of the following is true for a polytropic process? Explanation: These are the properties of an adiabatic process. Explanation: It comes from the p1*(v1^n) = p2*(v2^n).
What do you mean by thermodynamics process?
A thermodynamic process is defined as a change from one equilibrium macrostate to another macrostate. The initial and final states are the defining elements of the process. In this process energy may be transferred form or into the system and also work can be done by or on the system.
How is Polytropic process related to adiabatic process?
If the specific heat c of the polytropic change is zero, the polytropic process becomes an adiabatic one (dQ = 0). Also, if the specific heat of the polytrope is c = ±∞, the polytropic change becomes an isothermal one (dT = 0). Thus, polytropic processes are intermediate between isothermal and adiabatic processes.
What’s the difference between adiabatic and polytropic process?
The key difference between adiabatic and polytropic processes is that in adiabatic processes no heat transfer occurs whereas in polytropic processes heat transfer occurs. If the energy of a system changes because of a temperature difference, we say there has been a flow of heat.