What is a solid turning into a gas called?
What is a solid turning into a gas called?
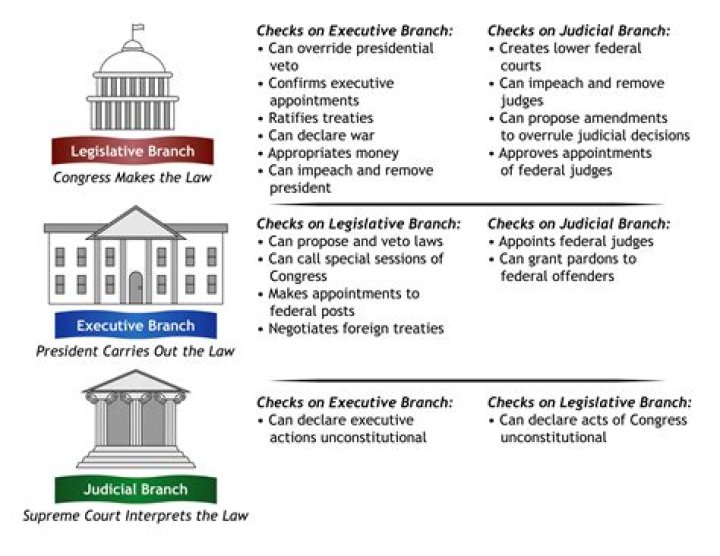
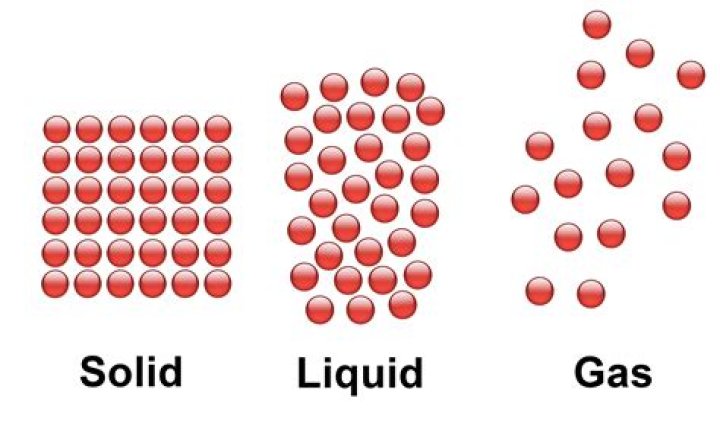
sublimation, in physics, conversion of a substance from the solid to the gaseous state without its becoming liquid. An example is the vaporization of frozen carbon dioxide (dry ice) at ordinary atmospheric pressure and temperature.
What is it called when solid water turns to gas?
This change, directly from a solid to a gas without becoming a liquid, is called sublimation. On crisp, dry winter days you might notice that snow banks shrink, or ice gradually disappears, without first becoming slushy and wet. When water absorbs enough heat, it becomes a gas (water vapor).
Which solid directly changes to gas?
– The process in which a solid directly gets converted directly to a gaseous state is called sublimation.
What is called sublimation?
Sublimation is the conversion between the solid and the gaseous phases of matter, with no intermediate liquid stage. The opposite of sublimation is “deposition”, where water vapor changes directly into ice—such a snowflakes and frost.
What is it called when something is a solid and liquid?
Solids and liquids are collectively called condensed phases because their particles are in virtual contact.
What is sublimation answer?
Sublimation is the process by which a substance changes from solid state directly to vapour state. Example : dry ice, naphthalene balls etc.
What is it called when a solid turns into a liquid?
The process of a solid becoming a liquid is called melting (an older term that you may see sometimes is fusion). For any pure substance, the temperature at which melting occurs — known as the melting point — is a characteristic of that substance. It requires energy for a solid to melt into a liquid.
What is an example of Desublimation?
Probably the most familiar example of desublimation is the formation of frost on a window in winter. Water vapor in cold air freezes into ice without ever becoming liquid water. This is also how hoar frost forms and accounts for some frost formation in home freezers. Another example is soot formation in chimneys.
What does it mean when a liquid turns into a gas?
evaporation
When a liquid changes into gas, it is called evaporation. Phase changes are usually caused by changes in temperature or pressure. When more heat is added, the liquid substance evaporates and becomes a gas, which has no bonds at all.
What process occurs when liquid turns into gas?
Evaporation happens when a liquid substance becomes a gas. When water is heated, it evaporates. The molecules move and vibrate so quickly that they escape into the atmosphere as molecules of water vapor.
Why do substances sublime?
Sublimation is an analogous process to boiling, as it occurs when a compound’s vapor pressure equals its applied pressure (often the atmospheric pressure). Most solids do not have an appreciable vapor pressure at easily accessible temperatures, and for this reason the ability to sublime is uncommon.