What is an even sharing of electrons called?
What is an even sharing of electrons called?
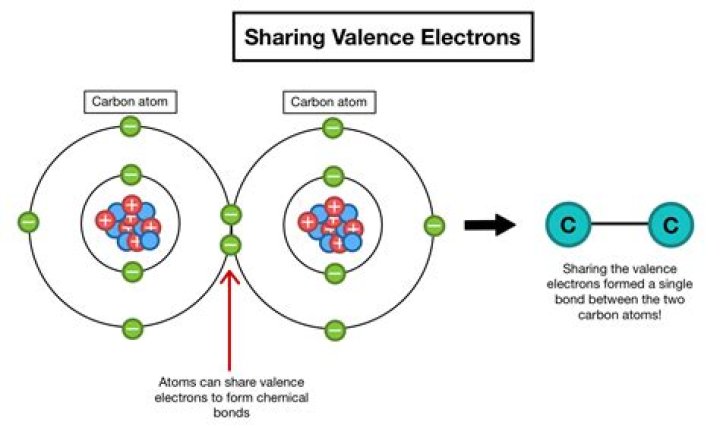
A covalent bond is a chemical bond that involves the sharing of electron pairs between atoms. These electron pairs are known as shared pairs or bonding pairs, and the stable balance of attractive and repulsive forces between atoms, when they share electrons, is known as covalent bonding.
What is the sharing of two electrons called?
When electrons are shared between two atoms, they make a bond called a covalent bond.
What is it called when electrons are shared unequally?
This arrangement, in which one atom in a covalent bond is slightly more negative and the other slightly more positive, creates a dipole. A bond in which electrons are shared unevenly is known as a polar bond.
Why are electrons shared unequally?
In pure covalent bonds, the electrons are shared equally. In polar covalent bonds, the electrons are shared unequally, as one atom exerts a stronger force of attraction on the electrons than the other. The difference in electronegativity between two atoms determines how polar a bond will be.
What are shared pairs?
Shared pair: A pair of electrons which is shared with other atoms to form a bond is known as shared pair of electrons. For example in HCl the pair of electrons responsible for bond formation between H and Cl is called shared pair.
What electrons are transferred?
The attraction between oppositely charged ions is called an ionic bond, and it is one of the main types of chemical bonds in chemistry. Ionic bonds are caused by electrons transferring from one atom to another.
What is the electronegativity difference of CO?
The electronegativity of carbon is about 2.5 , and that of oxygen is about 3.5 , and naturally, (3.5−2.5=1.0)>0 . So, the electronegativity difference is greater for a C−O single bond than a F−F single bond.
What causes unequal sharing of electrons?
Atoms with high electronegativity values—such as fluorine, oxygen, and nitrogen—exert a greater pull on electrons than do atoms with lower electronegativity values. In a bond, this can lead to unequal sharing of electrons between atoms, as electrons will be drawn closer to the atom with higher electronegativity.
What bond is electrons are shared unequally?
polar Covalent bonds
Covalent bonds involve the sharing of electrons between two atoms, but those electrons are not always shared equally. As the electronegativity difference between atoms in a covalent bond increases, electron sharing becomes less even.
Which of the following substances is formed by sharing electrons unequally?
2. Non-polar covalent bond. Polar covalent bond is formed by unequal sharing of electrons and in non polar covalent bonds number of electrons shared between two atoms are equal.
What does unequal sharing of electrons mean?
polar covalent bond
A polar covalent bond is a covalent bond in which the atoms have an unequal attraction for electrons and so the sharing is unequal. In a polar covalent bond, sometimes simply called a polar bond, the distribution of electrons around the molecule is no longer symmetrical.
What is lone pair of electrons?
In chemistry, a lone pair refers to a pair of valence electrons that are not shared with another atom in a covalent bond and is sometimes called an unshared pair or non-bonding pair. Lone pairs are found in the outermost electron shell of atoms. They can be identified by using a Lewis structure.