What is diazotization reaction of amine?
What is diazotization reaction of amine?
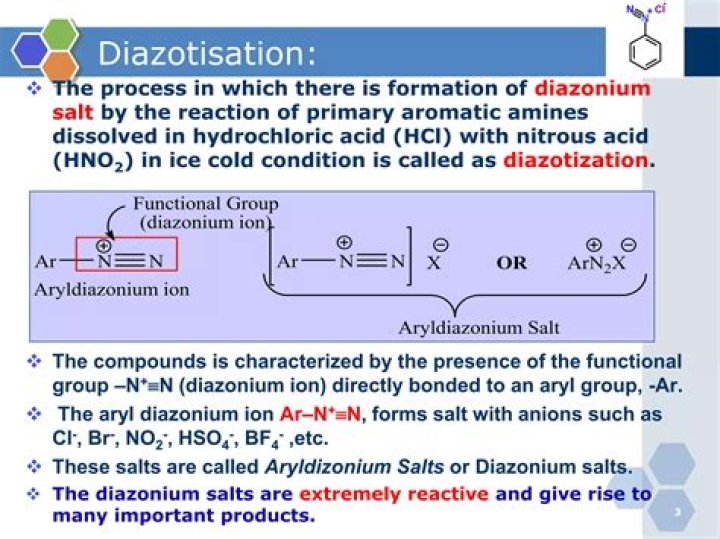
What is Diazotization Reaction? Aromatic amine reacts with nitrous acid and mineral acid to form diazonium salt and produces water as a side product. This reaction is known as Diazotization Reaction.
Which amines give diazotization reactions?
Diazotization is an important reaction of 1° amines. In the diazotization process, the NH2 group is changed to a diazonium salt, R–N2+X−. This is done by reaction with nitrous acid (HNO2). The reactive salt is not usually isolated.
What is diazotization reaction give example?
Aromatic primary amines reacts with nitrous acid in cold condition to give diazonium salts. Ph−NH2+NaNO2+2HCl273−278K PhN2+Cl−+NaCl+2H2O. Diazonium salts are stable for short time and decompose slowly even at low temperatures. They are used immediately after preparation.
What amines can form a diazonium salt?
The nitrosation of primary aromatic amines with nitrous acid (generated in situ from sodium nitrite and a strong acid, such as hydrochloric acid, sulfuric acid, or HBF4) leads to diazonium salts, which can be isolated if the counterion is non-nucleophilic.
What is diazotization reaction of aniline?
Much like in primary aliphatic amines, aminobenzene (aniline) is converted into a diazonium ion by treating it with a nitrous acid and mineral acid mixture. Formally, a nitrosyl cation () is then transferred to the amine.
What is diazotization write diazotization reaction for aniline?
Diazotization reaction involves the conversion or primary amines to the benzene diazonium salt. When aniline reacts with the sodium nitrite in the presence of an acid like hydrochloric acid at 273-278K, it leads to the formation of benzene diazonium salt. This reaction is generally performed by aromatic amines.
What is diazotization write diazotization reaction of aniline?
Solution 2 Diazotisation: The process of conversion of a primary aromatic amino compound into a diazonium salt, is known as diazotisation. This process is carried out by adding an aqueous solution of sodium nitrite to a solution of primary aromatic amine (e.g., aniline) in excess of HCl at a temperature below 5°C.
What is diazotization reaction Class 12?
Diazotization is a process of converting aromatic amine by nitrous acid below 5° to a diazonium salt. (ii) If the solution is turbid or ppt appears and remains unaffected by the addition of an acid, the given amine is a secondary amine.
What is diazotization Class 12?
It is a chemical process involved in organic chemistry. – It is the conversion of primary aromatic amine into the diazonium salt of amine by the use of nitrous acid; also known as diazotization.
Why HCl is used in Diazotisation?
In the diazotisation of aniline with sodium nitrite and hydrochloric acid, an excess of hydrochloric acid is used primarily to : Suppress the concentration of free aniline available for coupling. Suppress hydrolysis of phenol.
Which compound undergo diazotization most readily?
aryl amine
From the given aryl amine, the one that undergoes diazotization most readily, in the first option, the withdrawing nature of the NO₂ group leads to diazotization most readily.
How can you distinguish between hexylamine and aniline?
Ethylamine and aniline can bedistinguished from each other by the azo-dye test. A dye is obtained whenaniline (an aromatic amine) reacts with HNO2 (NaNO2 + dil. HCl) at 0-5°C, followed by a reaction with the alkaline solution of 2-naphthol.