What is elimination reaction and its mechanism?
What is elimination reaction and its mechanism?
An elimination reaction is a type of organic reaction in which two substituents are removed from a molecule in either a one- or two-step mechanism. The one-step mechanism is known as the E2 reaction, and the two-step mechanism is known as the E1 reaction.
What are the differences between E1 E2 and E1cB reactions?
Although E1 reactions typically involve a carbocation intermediate, the E1cB reaction utilizes a carbanion intermediate. This poor leaving group makes the direct E1 or E2 reactions difficult. This reaction is used later in a reaction called an aldol condensation.
What is E1cB mechanism in chemistry?
The E1cB (Elimination, Unimolecular, Conjugate Base) mechanism is a third mechanistic pathway for elimination reactions. In many ways it is the exact opposite of the E1 mechanism, as the first step is deprotonation to form a carbanion, followed by elimination in the second step.
What are the types of elimination reaction?
Types of Elimination Reaction
- E1 type. two-step removal mechanism process. also known as unimolecular elimination. formation of an intermediate.
- E2 type. one step removal mechanism process. also known as bimolecular reaction.
- E1cb type. two-step elimination mechanism. also known as unimolecular conjugate base elimination.
What is the difference between E1 and E2 mechanism?
The key differences between the E2 and E1 mechanism are: 1) E2 is a concerted mechanism where all the bonds are broken and formed in a single step. The E1, on the other hand, is a stepwise mechanism. 3) E2 is a second-order reaction and the rate depends on the concentration of both, the substrate and the base.
How do you know if it’s E1 or E2?
Comparing E1 and E2 mechanisms 1) The base: strong bases favor the E2 mechanism, whereas, E1 mechanisms only require a weak base. 2) The solvent: good ionizing xolvents (polar protic) favor the E1 mechanism by stabilizing the carbocation intermediate.
What is the difference between E2 and E1cB?
The key difference between the E2 vs E1cb pathways is a distinct carbanion intermediate as opposed to one concerted mechanism….Distinguishing E1cB-elimination reactions from E1- and E2-elimination reactions.
| E1 | E2 | E1cB |
|---|---|---|
| Stepwise reaction | Concerted reaction | Stepwise reaction |
Which of the following will give E1cB mechanism?
In 2-bromo-6-nitro cyclohexyl acetate gives the E1cB reaction.
What is the Hofmann product in an elimination reaction?
Hofmann elimination is an elimination reaction of an amine. The least stable alkene (the one with the least number of substituents on the carbons of the double bond), called the Hofmann product, is formed.
What are the two possible elimination mechanisms?
In this reaction, a substrate (typically an alkyl halide) eliminates one equivalent (unit) of acid to form an alkene. Two possible mechanisms are available for this elimination reaction – E1 and E2 mechanisms.
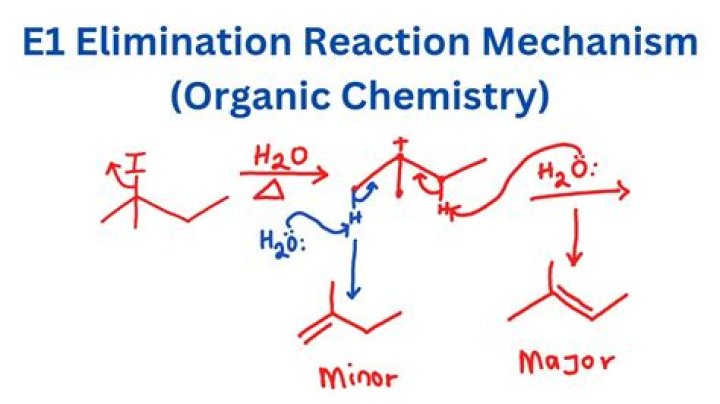
What is E1 reaction mechanism?
Unimolecular Elimination (E1) is a reaction in which the removal of an HX substituent results in the formation of a double bond. It is similar to a unimolecular nucleophilic substitution reaction (SN1) in various ways. One being the formation of a carbocation intermediate.
What is the difference between elimination and substitution reaction?
The difference between substitution and elimination reaction is shown below….Complete step by step answer:
| Substitution reaction | Elimination reaction |
|---|---|
| In this reaction, one atom or group is replaced by the other group or atom. | In this reaction, the atom or group is completely removed. |