What is Enediol reaction of carbohydrate?
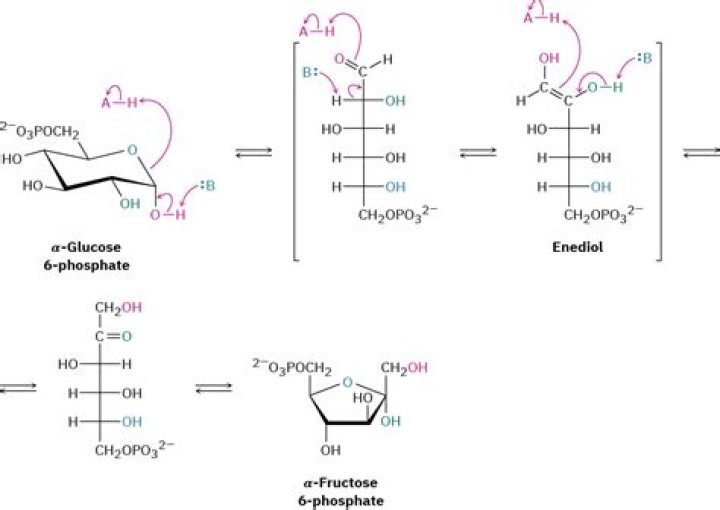
What is Enediol reaction of carbohydrate?
The enediol is also an intermediate for the epimerization of an aldose or ketose. The reactions are usually base catalyzed, but can also take place under acid or neutral conditions. A typical rearrangement reaction is that between the aldose glyceraldehyde and the ketose dihydroxyacetone in a chemical equilibrium.
What is Enediol rearrangement with example?
This transformation occurs at basic medium and allows the conversion of epimers, defined as isomeric forms that differ in the position of the hydroxyl group at C-2. Thus, the enolization proceeds by migration of proton at position 2, to carbon at 1 (Figure 1.13). …
Why Enediols are highly reactive?
In mildly alkaline solns, carbohydrates containing free aldehyde or keto group will tautomerise to form enediols (compounds where 2 –OH grp containing C-atoms are linked by double bonds). Enediols are highly reactive and capable of reducing ions. Eg., Cupric ions to cuprous ions.
Is Enediol a reducing agent?
Therefore, the chemical equilibrium produces mainly the enediol form rather than the keto form. Reductones are reducing agents, thus efficacious antioxidants. Some are fairly strong acids….Reductone.
| Examples of reductones | ||
|---|---|---|
| Tartronaldehyde | Reductic acid | Ascorbic acid (Vitamin C) |
What are epimers chemistry?
Epimers are diastereomers that contain more than one chiral center but differ from each other in the absolute configuration at only one chiral center. In epimers the chiral carbon atoms whose absolute configuration makes the two compounds different are called the epimeric carbons.
Can disaccharides be reducing sugars?
Disaccharides are formed from two monosaccharides and can be classified as either reducing or nonreducing. Reducing disaccharides like lactose and maltose have only one of their two anomeric carbons involved in the glycosidic bond, while the other is free and can convert to an open-chain form with an aldehyde group.
Why are Enediols reducing agents?
The enediol structure is stabilized by the resonance resulting from the tautomerism with the adjacent carbonyl. Therefore, the chemical equilibrium produces mainly the enediol form rather than the keto form. Reductones are reducing agents, thus efficacious antioxidants.
What is the term used to describe carbohydrates that can be oxidized by Benedict’s reagent?
Reducing Sugar. What is the term used to describe carbohydrates that can be oxidized by Benedict’s reagent? -All monosaccharides and Disaccharides (Expect Sucrose) Ans: Benedict’s test.
Why ketoses are reducing sugars?
All monosaccharide ketoses are reducing sugars, because they can tautomerize into aldoses via an enediol intermediate, and the resulting aldehyde group can be oxidised, for example in the Tollens’ test or Benedict’s test.
How do you identify epimers?
An epimer is one of a pair of stereoisomers that differ in configuration at only one chiral (stereogenic) centre.
- An epimer is one of a pair of stereoisomers that differ in configuration only at the chiral centre.
- All other stereocenters (if there are any) are the same in both molecules.
How are epimers formed?
7.1. Epimerization is a process in stereochemistry in which there is a change in the configuration of only one chiral center. As a result, a diastereomer is formed. The classical example of this in medicine is tetracycline.