What is Group 6 on the periodic table?
What is Group 6 on the periodic table?
Group 6, numbered by IUPAC style, is a group of elements in the periodic table. Its members are chromium (Cr), molybdenum (Mo), tungsten (W), and seaborgium (Sg). These are all transition metals and chromium, molybdenum and tungsten are refractory metals.
What is number 6 on the periodic table of elements?
Carbon
Carbon the element that is atomic number 6 on the periodic table.
What properties do Group 6 elements have?
O, S, Se, Te and Po are the elements of group VI-A.
What element is found in Group 6 and Period 4?
Chromium (Cr) is an element in group 6.
What is Group 6 Period 6 on the periodic table?
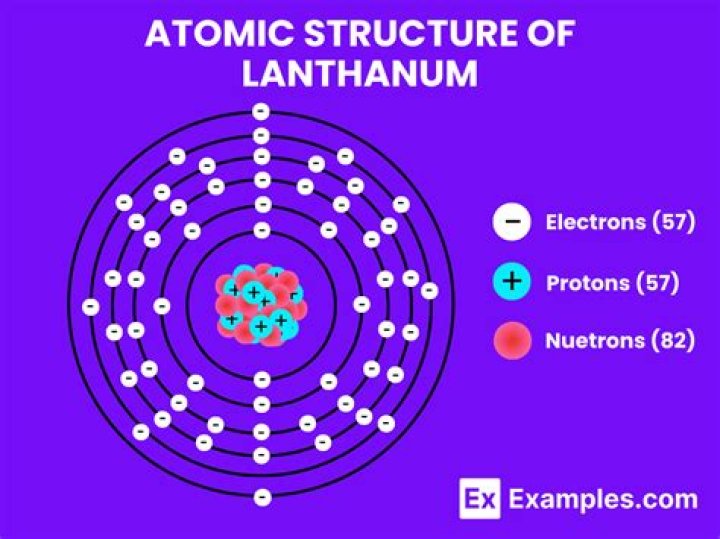
The period 6 transition metals are lanthanum (La), hafnium (Hf), tantalum (Ta), tungsten (W), rhenium (Re), osmium (Os), iridium (Ir), platinum (Pt), gold (Au), and mercury (Hg).
Which element in Group 6 is least reactive?
Noble Gases
Chemical Properties of Noble Gases Noble gases are the least reactive of all known elements. Their outer energy levels are full because they each have eight valence electrons. The only exception is helium, which has just two electrons.
What is the 6 in Carbon?
The element carbon (C) has an atomic number of 6, which means that all neutral carbon atoms contain 6 protons and 6 electrons. In a typical sample of carbon-containing material, 98.89% of the carbon atoms also contain 6 neutrons, so each has a mass number of 12.
What is the atomic number of 6?
Carbon is a chemical element. Its atomic number is 6; its atomic weight is 12.011.
Which element in group 6 is least reactive?
Is Cl2 a halogen?
The group of halogens is the only periodic group that contains elements in three of the main states of matter at standard temperature and pressure. Fluorine and chlorine are gases, bromine is liquid and iodine and astatine are solid. Halogens form diatomic molecules such as F2, Cl2, Br2 or I2 in their elemental states.
What element is in Period 6 Group 18?
Period 6
| Group | 1 | 18 |
|---|---|---|
| Atomic # Name | 55 Cs | 86 Rn |
Why is chromium in group 6?
chromium (Cr), chemical element of Group 6 (VIb) of the periodic table, a hard steel-gray metal that takes a high polish and is used in alloys to increase strength and corrosion resistance….chromium.
| atomic number | 24 |
|---|---|
| specific gravity | 7.20 (28 °C) |
| oxidation states | +2, +3, +6 |
| electron configuration | [Ar]3d54s1 |