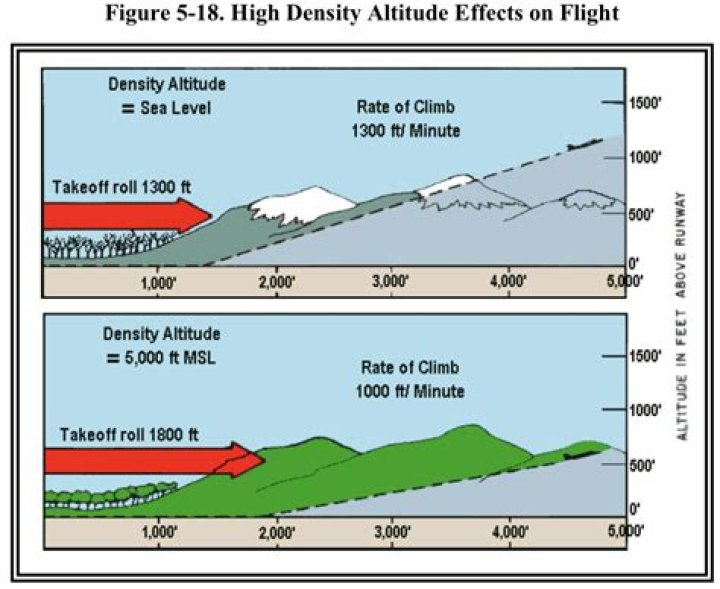
What is interchangeable drug?
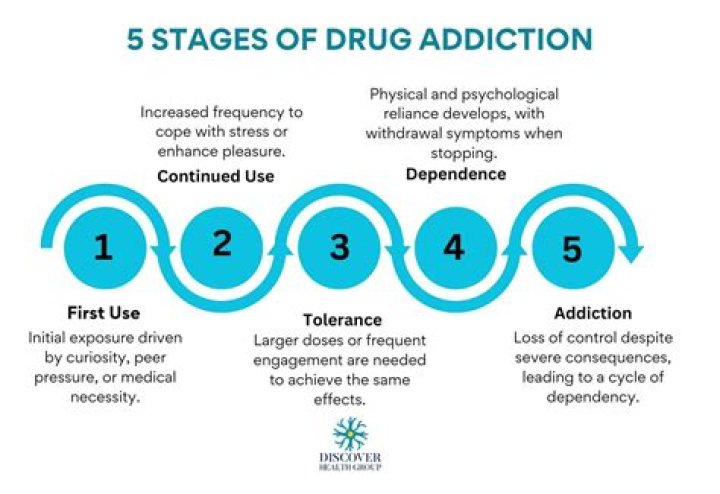
What is interchangeable drug?
“Interchangeable drug” means a drug that: * contains the same amount of the same active ingredients, * possesses comparable pharmacokinetic properties, * has the same clinically significant formulation characteristics,1 and * is to be administered in the same way as the drug prescribed.
What is an interchangeable product?
What is an interchangeable product? An interchangeable product is a biosimilar product that meets additional requirements outlined by the Biologics Price Competition and Innovation Act.
What is the difference between biosimilar and interchangeable?
Interchangeable biologics are biosimilars that are approved by the FDA to be substituted by a third party for the reference biologic. An interchangeable biologic is not superior in quality to a biosimilar product as both have to meet the very same high standards.
What is product interchangeability in pharmaceutical?
An interchangeable pharmaceutical product is one which is therapeutically equivalent to a comparator product and can be interchanged in clinical practice.
What is a biologic Med?
Biologic medications are drugs that target specific parts of your immune system to treat disease. If you have a condition like psoriatic arthritis, biologics can make a big difference. They can ease inflammation in your body, stop joint damage, and improve your quality of life.
What are interchangeable biosimilar medications?
An interchangeable biosimilar product may be substituted without the intervention of the health care professional who prescribed the reference product, much like how generic drugs are routinely substituted for brand name drugs. This is commonly called pharmacy-level substitution and is subject to state pharmacy laws.
What is a biologic in medicine?
Biologics are powerful medications that can be made of tiny components like sugars, proteins, or DNA or can be whole cells or tissues. These drugs also come from all sorts of living sources — mammals, birds, insects, plants, and even bacteria.
What is the difference between biologic and biosimilar drugs?
Biologic drugs are large, complex proteins made from living cells through highly complex manufacturing processes. Unlike generic drugs, which are copies of chemical drugs, a biosimilar is a copy of a biologic medicine that is similar, but not identical, to the original medicine.
Is Humira a biologic or biosimilar?
Humira is the brand name for adalimumab, a biologic approved by the FDA in 2002. In 2016, the FDA approved a biosimilar for Humira called Amjevita.
What is interchangeability FDA?
Interchangeable Biological Products. An interchangeable biological product is a biosimilar that meets additional requirements and may be substituted for the reference product at the pharmacy, depending on state pharmacy laws.
What is the difference between a drug and a biologic?
A biologic is manufactured in a living system such as a microorganism, or plant or animal cells. A drug is typically manufactured through chemical synthesis, which means that it is made by combining specific chemical ingredients in an ordered process.