What is isotropic liquid?
What is isotropic liquid?
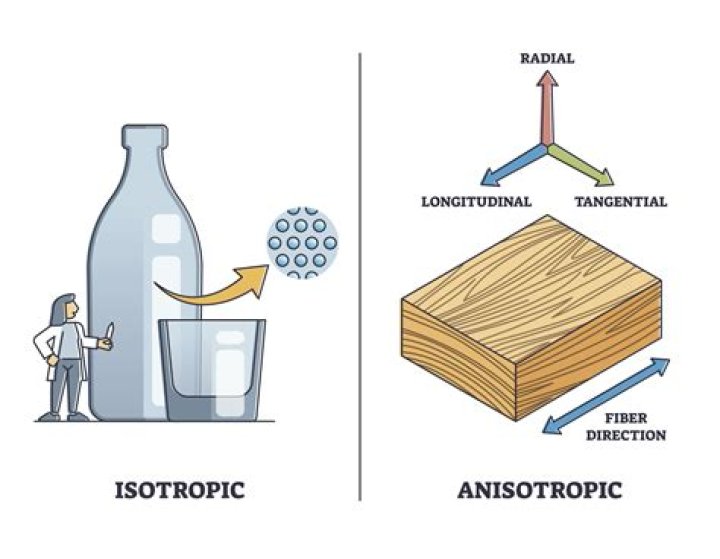
A true liquid is isotropic , meaning that its properties are uniform in all directions— the result of its molecules being in constant random motion. Crystalline solids, in contrast, are anisotropic ; optical- and other properties such as thermal and electrical conductivity vary with direction.
What does isotropic mean in chemistry?
Isotropy is a common term in materials science that means uniform in all directions. Isotropic materials exhibit the same material properties in all directions. Metals and glasses tend to be isotropic. On the other hand, anisotropic materials exhibit different material properties in different directions.
What do you mean by Isotropy?
Isotropy is uniformity in all orientations; it is derived from the Greek isos (ἴσος, “equal”) and tropos (τρόπος, “way”). Isotropic radiation has the same intensity regardless of the direction of measurement, and an isotropic field exerts the same action regardless of how the test particle is oriented.
What are the types of mesophase of liquid crystal?
In calamitic mesogens, there are three types of mesophases namely the nematic, cholesteric and smectic originally classified by Friedel [7] based on the degree of positional and orientational order. The discovery of new phases in liquid crystals turns to be often associated with an increase of complexity.
Is liquid crystal isotropic?
Liquid crystals (LCs) are anisotropic fluids, thermodynamically situated between normal isotropic liquid and three-dimensionally ordered crystals.
What is anisotropy example?
anisotropic: Properties of a material depend on the direction; for example, wood. In a piece of wood, you can see lines going in one direction; this direction is referred to as “with the grain”. Strength is a property of the wood and this property depends on the direction; thus it is anisotropic.
What is isotropic point?
An isotropic radiator is a theoretical point source of electromagnetic or sound waves which radiates the same intensity of radiation in all directions. It has no preferred direction of radiation. It radiates uniformly in all directions over a sphere centred on the source.
What is nematic and smectic liquid crystals?
The key difference between nematic smectic and cholesteric liquid crystals is that nematic liquid crystals have no ordered structure of molecules, and smectic liquid crystals have a layered molecular structure, whereas cholesteric liquid crystals have molecules in a twisted and chiral arrangement.
What is the difference between thermotropic and lyotropic liquid crystals?
Thermotropic liquid crystalline phases are formed by a change of temperature, whereas lyotropic phases are formed when mixed with aqueous phase. The phase transitions of thermotropic liquid crystal are temperature-dependent, while those of lyotropic liquid crystals depend on both temperature and concentration [5].