What is meant by singlet and triplet state?
What is meant by singlet and triplet state?
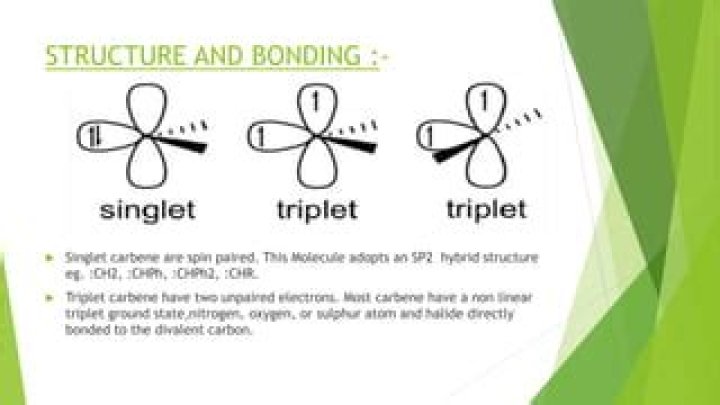
Singlet state: All electrons in the molecule are spin paired. It is called a singlet because there is only one possible orientation in space. Triplet state: One set of electron spins is unpaired. It is called a triplet because there are three possible orientations in space with respect to the axis.
What is singlet doublet and triplet state?
. As a result, there is only one spectral line of a singlet state. In contrast, a doublet state contains one unpaired electron and shows splitting of spectral lines into a doublet; and a triplet state has two unpaired electrons and shows threefold splitting of spectral lines.
Why is it called triplet state?
A triplet state is an electronic state in which two electrons in different molecular orbitals have parallel spins, as shown in Fig. The name “triplet” reflects that there are three triplet sublevels as discussed earlier (see Section 4.15. 1). These sublevels are degenerate only for spherical molecular symmetry.
What is the difference between triplet oxygen and singlet oxygen?
The singlet has only one possible arrangement of electron spins with a total quantum spin of 0, while the triplet has three possible arrangements of electron spins with a total quantum spin of 1, corresponding to three degenerate states.
What is a singlet representation?
A singlet of any algebra is the state that has zero eigenvalue under the Casimir operators. These are the generators of the center of the algebra and are simultaneously diagonalizable. The singlet state is a one-dimensional representation. For instance, for , the singlet state satisfies. i.e., it is the state.
What is a triplet state in physics?
In quantum mechanics, a triplet is a quantum state of a system with a spin of quantum number s=1, such that there are three allowed values of the spin component, ms = −1, 0, and +1.
What is singlet and doublet?
Doublet states are from single spins. Triplet and singlet states are from pairs of spins. The triplet and singlet states diagonalize S^2, with S being the total spin of the system.
Is singlet state Antisymmetric?
Thus the singlet state is antisymmetric and the triplet state is symmetric when exchanging the indices. Always the complete wave function of the two electron state has to be antisymmetric; consequently the ”local” wave function of a singlet state must be symmetric and for a triplet state it has to be antisymmetric.
Where does the singlet state come from?
A singlet state is formed when a molecular configuration has no net spin (ζ=0), i.e. for every electron that has spin ½ (up), there is an electron that has spin−½ (down).
Why is it called singlet and triplet?
It’s called the “singlet” because there is only one state with total spin, 0. The triplet is the set of states with total spin equal to 1: Again, it’s called the “triplet,” because there are three states with total spin equal to 1.
What are the main differences between singlet and triplet excited state of organic molecules?
In an excited singlet state, the electron is promoted in the same spin orientation as it was in the ground state (paired). In a triplet excited stated, the electron that is promoted has the same spin orientation (parallel) to the other unpaired electron.
Why singlet state is more energy than triplet state?
Because the singlet state has electron repulsion and no exchange energy, i.e. it has paired antiparallel electrons instead of unpaired parallel electrons. Triplet: two unpaired electrons.