What is tetrahedron in chemistry?
What is tetrahedron in chemistry?
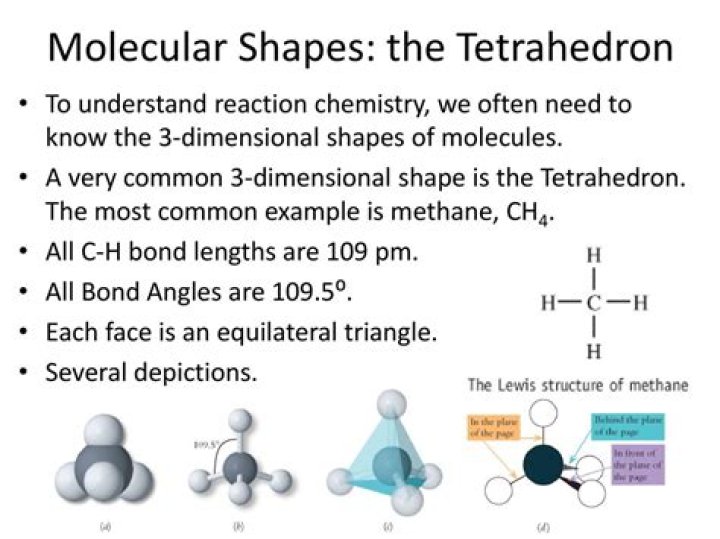
Tetrahedral is a molecular shape that results when there are four bonds and no lone pairs around the central atom in the molecule. The atoms bonded to the central atom lie at the corners of a tetrahedron with 109.5° angles between them.
What makes a molecule tetrahedral?
In a tetrahedral molecule, there is one central atom bonded to four surrounding atoms with no lone electron pairs. The bonds form angles of 109.5°. Some examples of tetrahedral molecules include the ammonium ion, methane ion, and phosphate ion.
What is the tetrahedral shape?
Tetrahedral. Tetra- signifies four, and -hedral relates to a face of a solid; “tetrahedral” literally means “having four faces. ” This shape is found when there are four bonds all on one central atom, with no lone electron pairs. In accordance with the VSEPR theory, the bond angles between the electron bonds are 109.5o …
What molecules have a tetrahedral shape?
Tetrahedral. Molecules of methane, CH4, ammonia, NH3, and water, H2O, all have four electron groups around their central atom, so they all have a tetrahedral shape and bond angles of about 109.5°.
What is linear structure in chemistry?
Linear Structure Definition The linear molecular geometry in chemistry explains the structure surrounding a central atom bound to two other atoms (or ligands) positioned at a 180° bond angle.
Is a triangular pyramid a tetrahedron?
In geometry, a tetrahedron (plural: tetrahedra or tetrahedrons), also known as a triangular pyramid, is a polyhedron composed of four triangular faces, six straight edges, and four vertex corners.
What is a linear shape?
Linear = is just a line of atoms with a 180° angle. Notice that it’s 2 or 3 atoms total. Bent = Linear but bent due to the Lone Pairs that it contains, the more Lone Pairs the greater the bent and the smaller the degree.
What molecules are linear?
Linear molecule is a molecule in which atoms are deployed in a straight line (under 180° angle). Molecules with an linear electron pair geometries have sp hybridization at the central atom. An example of linear electron pair and molecular geometry are carbon dioxide (O=C=O) and beryllium hydride BeH2.
What is pyramidal shape?
A pyramid is a polyhedron for which the base is a polygon and all lateral faces are triangles. Technically, when the lateral faces are congruent triangles, the shape is known as a right pyramid, indicating that the apex — the vertex at which the lateral faces meet — is directly above the center of the base. …
Does nh3 have a tetrahedral shape?
Ammonia has 4 regions of electron density around the central nitrogen atom (3 bonds and one lone pair). These are arranged in a tetrahedral shape. The resulting molecular shape is trigonal pyramidal with H-N-H angles of 106.7°.
How do you find the linear structure?
The number of bonded pairs is equal to the number of substrates attached to the central element of the binary compound and represents the number of ‘Bonded Pairs’ of electrons. #BPrs = 2. => AX2 => Linear Geometry X – A – X for the BeCl2 structure. That is, Cl – Be – Cl is a linear molecule.
Is tetrahedron a prism or pyramid?
The tetrahedron is one kind of pyramid, which is a polyhedron with a flat polygon base and triangular faces connecting the base to a common point. In the case of a tetrahedron the base is a triangle (any of the four faces can be considered the base), so a tetrahedron is also known as a “triangular pyramid”.