What is the antigen binding site on the antibody
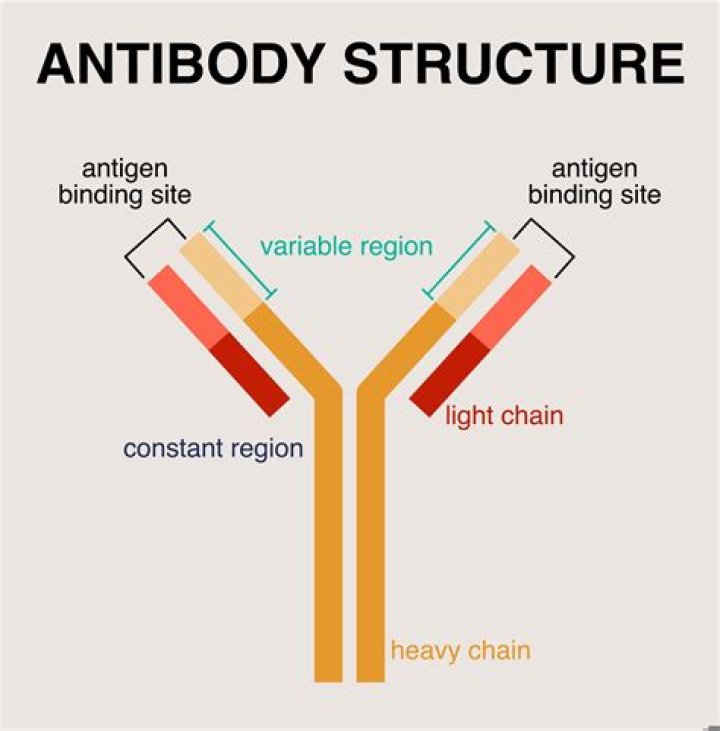
The paratope is the part of an antibody which recognizes an antigen, the antigen-binding site of an antibody. It is a small region (15–22 amino acids) of the antibody’s Fv region and contains parts of the antibody’s heavy and light chains. The part of the antigen to which the paratope binds is called an epitope.
What is the antigen binding site?
(A) The hinge region of an antibody molecule opens and closes to allow better binding between the antibody and antigenic determinants on the surface of an antigen. (B) Hinge flexibility also facilitates the cross-linking of antigens into large antigen-antibody complexes.
Where is the antigen binding site of this antibody quizlet?
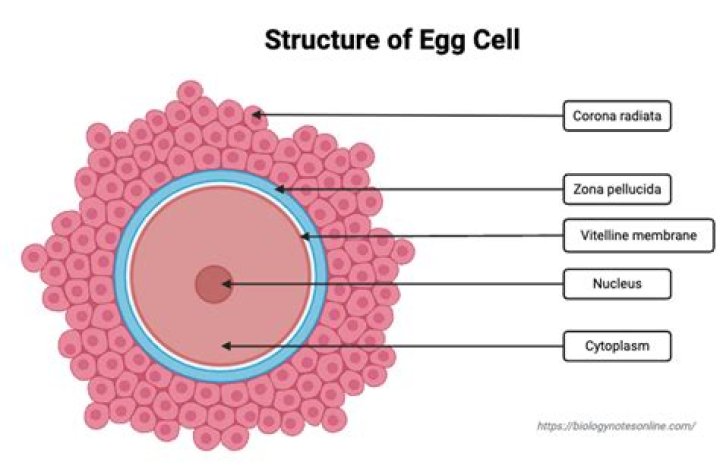
The antigen binding site is made up of the combined variable regions of the light and heavy chains. This can be called the Fab region, and there are 2/antibody. What determines the Fc region of the antibody? The Fc region is determined by the constant region of the antibody’s genome.
Why are there two antigen binding sites?
The possession of two antigen-binding sites allows antibody molecules to cross-link antigens and to bind them much more stably. The trunk of the Y, or Fc fragment, is composed of the carboxy-terminal domains of the heavy chains. Joining the arms of the Y to the trunk are the flexible hinge regions.What is the antibody binding site made up of?
It is composed of four polypeptide chains—two identical heavy chains and two identical light chains. The two antigen-binding sites are identical, each formed by the N-terminal region of a light (more…)
What does the word epitopes mean?
Definition of epitope : a molecular region on the surface of an antigen capable of eliciting an immune response and of combining with the specific antibody produced by such a response. — called also determinant, antigenic determinant.
How many antigen binding sites are in IgG?
IgG is the most common class of immunoglobulin. It is present in the largest amounts in blood and tissue fluids. Each IgG molecule consists of the basic four-chain immunoglobulin structure—two identical H chains and two identical L chains (either kappa or lambda)—and thus carries two identical antigen-binding sites.
What is the purpose of antibodies binding to antigens?
Antibodies recognize foreign invading microorganisms by specifically binding to a pathogen’s proteins or antigens, facilitating their neutralization and destruction. Antigens are classically defined as any foreign substance that elicits an immune response.How is the antigen binding site formed?
The surface of the antibody molecule formed by the juxtaposition of the CDRs of the heavy and light chains creates the site to which an antigen binds.
How many antigen binding sites does IGA have?Each Ig monomer contains two antigen-binding sites and is said to be bivalent. The hinge region is the area of the H chains between the first and second C region domains and is held together by disulfide bonds.
Article first time published onHow many antigen binding sites does an immunoglobulin have quizlet?
There are two light chains and two heavy chains. The variable (Fab) region forms the antigen binding site. There is a variable region on both the heavy and light chain. There are two antigen binding sites on each antibody.
What does the constant region of an antibody do?
The constant region determines the mechanism used to destroy antigen. Antibodies are divided into five major classes, IgM, IgG, Iga, IgD, and IgE, based on their constant region structure and immune function.
What region of the antibody determines the particular class to which an antibody belongs?
The epitope is the part of the antigen that is recognized by the antibody. In addition, the constant regions of the heavy chains determine the immunoglobulin class to which the antibody belongs ( IgM , IgG , IgD , IgA or IgE ). The immunoglobulin class is also called isotype.
On what portion of an antigen do the antibodies bind quizlet?
Epitope is the small region on an antigen that binds to the variable region of an antibody molecule.
What is epitope and Paratope?
An epitope, also known as antigenic determinant, is the part of an antigen that is recognized by the immune system, specifically by antibodies, B cells, or T cells. The epitope is the specific piece of the antigen to which an antibody binds. The part of an antibody that binds to the epitope is called a paratope.
What does binding to an antigen mean?
Antigen-binding molecules. Antigen-binding molecules consist of three examples, which result in the specificity of the acquired immune response. They are able to bind to foreign antigens, and include the immunoglobulin, T-cell receptor, and major histocompatibility complex molecules.
Where is IgG found?
Immunoglobulin G (IgG), the most abundant type of antibody, is found in all body fluids and protects against bacterial and viral infections. Immunoglobulin M (IgM), which is found mainly in the blood and lymph fluid, is the first antibody to be made by the body to fight a new infection.
What are antigen presenting cells?
Antigen-presenting cells (APC) are cells that can process a protein antigen, break it into peptides, and present it in conjunction with class II MHC molecules on the cell surface where it may interact with appropriate T cell receptors.
What is the difference between antigen and epitope?
An epitope (also known as the antigenic determinant) is that part of the antigen to which antibodies bind. While the antigen evokes the antibody response in the host, the antibody doesn’t bind to the entire protein, but only to that segment called the epitope.
Is IgG monoclonal or polyclonal?
Polyclonal antibodies contain a heterologous mixture of IgGs against the whole antigen, whereas monoclonal antibodies are composed of a single IgG against one epitope (Figure 1.)
What is hapten immunology?
Haptens are small molecules that elicit an immune response only when attached to a large carrier such as a protein; the carrier may be one that also does not elicit an immune response by itself (in general, only large molecules, infectious agents, or insoluble foreign matter can elicit an immune response in the body).
Where are antibodies located?
Antibodies and immunoglobulins Immunoglobulins are found in blood and other tissues and fluids. They are made by the plasma cells that are derived from the B cells of the immune system. B cells of the immune system become plasma cells when activated by the binding of a specific antigen on its antibody surfaces.
Where are antigens found?
They recognise foreign substances, such as germs, and alert your immune system, which destroys them. Antigens are protein molecules found on the surface of red blood cells.
What does IgA bind to?
Secretory IgA Polymeric IgA (mainly the secretory dimer) is produced by plasma cells in the lamina propria adjacent to mucosal surfaces. It binds to the pIgR on the basolateral surface of epithelial cells, and is taken up into the cell via endocytosis.
Why is IgA a dimer?
Secretory IgA, a dimer, provides the primary defense mechanism against some local infections because of its abundance in mucosal secretions (e.g., saliva and tears). The principal function of secretory IgA may be not to destroy antigens but to prevent passage of foreign substances into the circulatory system.
Where is IgA from?
In most higher vertebrates, the majority of IgA is synthesized by gastrointestinal lymphoid tissue, with smaller amounts synthesized at other mucosal sites such as the respiratory tract, salivary glands and reproductive tract. During lactation, mammary tissue contains substantial numbers of IgA-producing cells.
How many antigen-binding sites are present on an IgG molecule quizlet?
IgM has ten antigen-binding sites per molecule, whereas IgG only has two.
How many antigen-binding sites does a typical IgM molecule have quizlet?
IgM – Pentamer , 5 chains, 10 binding sites .
What does IgA antibody do?
IgA antibodies protect body surfaces that are exposed to outside foreign substances. This type of antibody is also found in saliva, tears, and blood. About 10% to 15% of the antibodies present in the body are IgA antibodies. A small number of people do not make IgA antibodies.
What's the function of C region of IgG?
Since the mid-twentieth century, the Ig molecule has been considered a bifunctional molecule consisting of two largely independent regions, a V region responsible for specificity and affinity, and a C region responsible for effector functions such as complement activation and interaction with FcRs.
Why is it called the constant region?
composed of two regions, called constant (C) and variable (V). These regions are distinguished on the basis of amino acid similarity—that is, constant regions have essentially the same amino acid sequence in all antibody molecules of the same class (IgG, IgM, IgA, IgD, or IgE), but the amino acid sequences…