What is the chemical formula of sulfanilamide?
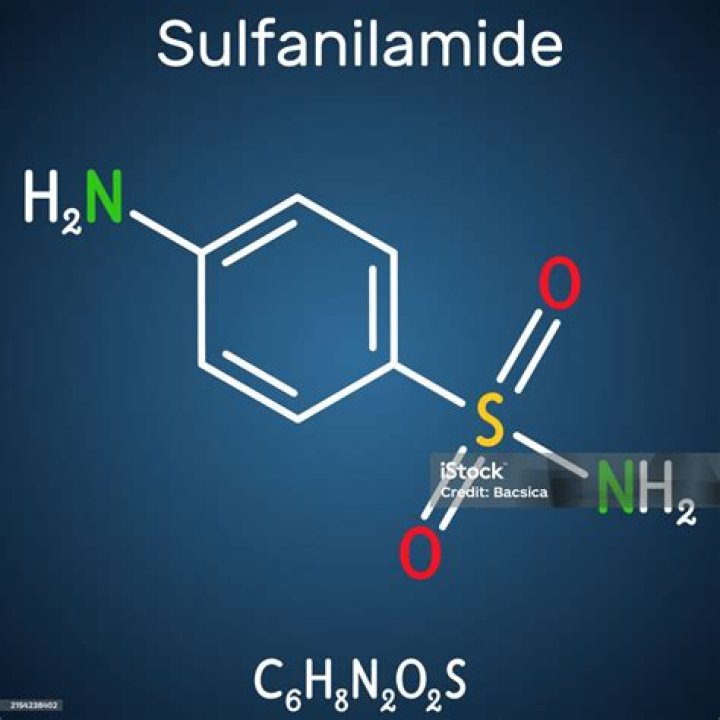
What is the chemical formula of sulfanilamide?
C6H8N2O2S
Sulfanilamide/Formula
Which one is the benzene sulfonamide derivative?
Celecoxib is a nonarylamine benzenesulfonamide derivative that was reported to have a very low potential for hepatotoxicity based on clinical trials with exposure of up to 2 years [241].
Which group is present in Sulphanilamide?
Sulfanilamide is a sulfonamide in which the sulfamoyl functional group is attached to aniline at the 4-position. It has a role as an EC 4.2.
Which compound is used as starting material in synthesis of sulfanilamide Mcq?
Sulphanilamide can beprepared by taking acetanilide and treating it with excess of chlorosulphonic acid, which gives p-acetamidobenzenesulphonyl chloride which readily converted into corresponding p-acetamidobenzenesulphonamides upon reaction with ammonia or ammonium carbonate.
What are Mech of action of sulfanilamide?
Mechanism of action As a sulfonamide antibiotic, sulfanilamide functions by competitively inhibiting (that is, by acting as a substrate analogue) enzymatic reactions involving para-aminobenzoic acid (PABA). Specifically, it competitively inhibits the enzyme dihydropteroate synthase.
What are Sulfanilamides analogous to?
The sulfonamides are a group of synthetic antimicrobial agents that are structural analogs of para-aminobenzoic acid (PABA).
Which sulfonamide does not contain free aromatic amino group?
There are structural differences between sulfonamide antibiotics and nonantibiotic sulfonamides (such as diuretics, oral hypoglycemics, carbonic anhydrase inhibitors, celecoxib and sumatriptan), in that the latter lack an N4 aromatic amine and an N1 substituted ring.
How do you make sulfa drugs?
Preparation. Sulfonamides are prepared by the reaction of a sulfonyl chloride with ammonia or an amine. Certain sulfonamides (sulfadiazine or sulfamethoxazole) are sometimes mixed with the drug trimethoprim, which acts against dihydrofolate reductase.
Is sulfapyridine a sulfonamide antibiotic?
Sulfapyridine is a short-acting sulfonamide antibiotic and by-product of the non-steroidal anti-inflammatory drug sulfasalazine. Its manufacture and use were discontinued in 1990. Sulfapyridine is a sulfonamide consisting of pyridine with a 4-aminobenzenesulfonamido group at the 2-position.
Is Prontosil still used today?
Prontosil has been replaced in clinical use by newer sulfonamide drugs, including sulfanilamide, sulfathiazole, sulfamethoxazole, and others.
When Acetanilide is treated with Chlorosulphonic acid what does it give?
This is the first step of the processes where required amount of acetanilide is reacted with chlorosulfonic acid to obtain an intermediate called p-acetamidobenzenesulfonyl chloride. This mixture is made to react in a reflux condenser for about 15-20 min where a semisolid paste is obtained which is brown.
What drugs are antibacterial drugs?
These approaches have also led to important new classes of antibacterial agents and large numbers of approved drugs. The most appreciated examples of synthetic substances used in therapeutics are the chloramphenicol, metronidazole, fosfomycin, quinolones, carbapenems, and oxazolidinones.